Colloids find many applications in our daily life and in industry. Some typical applications of colloids are given below:
1) Food
- Many constituents of our food are colloidal in nature. For example, milk, starch, proteins, fruit-jellies etc., are colloids.
2) Medicine
- A large number of medicines and pharmaceuticals preparations are colloidal in nature.
- Some of them are in fact emulsions. Such colloidal medicines are adsorbed by the body tissues and therefore are more effective.
- For example: colloidal god, calcium, silver are used in medicines or as ointments.
3) Sewage disposal
- The sewage contains fine particles of dirt and dust. These fine particles carry electrical charge.
- These fine colloidal particles can be coagulated (precipitated) by the application of electrical field.
- The sewage disposal tanks are fitted with metallic electrodes.
- The dirt particles move to the oppositely charged electrodes, get discharged and are precipitated.
4) Water treatment
- Raw water from rivers, lakes and canals contains many suspended and soluble impurities.
- The suspended mater is generally in the colloidal form. This matter cannot be separated by sedimentation and filtration.
- In such cases, alum and a little lime is added into the water to form precipitate of aluminium hydroxide.
- These precipitates adsorb the finer particles of the suspended material and thus help them in settling.
- The presence of Al3+ ions in solution also neutralizes the negative charge on the suspended sol particles and causes their coagulation.
- Thus, the suspended impurities are made to settle down and the clear water is decanted off.
5) Dyeing
- Dyeing involves adsorption of dye particles (molecules) on the surface of the fabric.
- Dyes are generally dissolved in suitable solvents to form a very fine suspension (colloidal).
- This increases the adsorption characteristics of the dye.
- In certain cases (mordant dyes), the dyes are adsorbed on the colloidal particles of the mordant.
6) Leather tanning
- Raw skin and hides contain large molecules in the form of cross-linked fibres.
- These protein macromolecules are positively charged. The tanning materials which include tannin and compounds of aluminium and chromium contain negatively charged sol particles.
- On soaking hides in such solutions, coagulation of negatively charged sol particles takes place in the pores of hide.
- This hardens the leather. Such hardening of leather is called tanning of leather.
7) Rubber industry
- Rubber obtained from the trees is an emulsion (called latex).
- Latex contains negatively charged rubber particles in water. On boiling, the protective layer of proteinous matter is broken and the rubber particles get coagulated.
- The coagulated mass is vulcanized by heating with sulphur and sold as rubber.
- Various articles are rubber-plated by electrolytic process.
- The article to be plated is made anode in the plating bath where negatively charged rubber particles get deposited.
8) Agriculture
- Soil is a mixture of clay, organic materials and various salts. The soil-clay is involved in many reactions e.g., ion-exchange reactions.
- To work out the treatment to be given to any soil, it is important to study its electrical/chemical behavior. Colloid chemistry provides such information.
- Many pesticides formulations are emulsions. Their preparation and storage is essentially based on the study of colloids.
9) Artificial rain
- Clouds consist of water vapours mixed with the particles of carbon/dust.
- The water molecules in clouds develop some electrical charge.
- These water molecules can be coagulated and precipitated by spraying fine materials such as sand, which neutralize the electrical charge on the water molecules and cause their coagulation.
10) Formation of deltas
- Extensive deposits of silt and clay formed in the sea at the mouth of the river is called delta.
- The charge on the clay particles suspended in the river water is neutralized by the salts present in the sea water. This causes coagulation of the clay.
11) Chemical warfare
- Smoke screens are used in wars to hide the movements of troops.
- Smoke screens generally consist of very fine particles of titanium oxide dispersed in air with the help of aircrafts.
- Titanium oxide screens are white opaque covers.
12) Cleansing actions of soaps and detergents
- Soaps and detergents remove oil/greasy dust from the soiled fabric by forming water-soluble emulsions through adsorption.
13) Smoke precipitations
- Smoke is a dispersion of fine dust and carbon particles in the air.
- The industrial waste gases containing suspended impurities make smoke more polluting.
- This leads to the pollution of air. These dispersions of suspended particles of colloidal dimensions are electrically charged.
- So, these can be removed from the air by electrical precipitation.
- To achieve this, a high voltage smoke precipitator, called Cottrell precipitator is used.
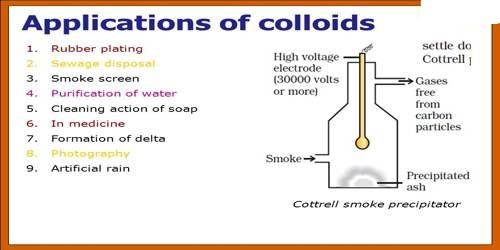
- The smoke along with gases and dust is led through a tower-like structure, fitted with a high voltage rod-like filament.
- The suspended particles get discharged when come in contact with the filament, and fall down to the bottom.
- The cleaner hot air leaves the precipitator from an exit near at the top.
- The Cottrell precipitators are being employed in coal based power generating units to reduce air pollution.
References:
ii) https://chemistry-desk.blogspot.com/2013/08/application-of-colloids.html

