- The atomic imperfections (defects) are caused by some disorder in the arrangement of the constituent particles in any crystal.
- There are two kinds of atomic defects. They are
i) Point defects
ii) Lattice defects
Point defects
- The defects caused by a departure from the periodic arrangement of particles in the vicinity of an atom or group of atoms, are called point defects.
- These defects involve only a few lattice points.
- The point defects occur in many ways.
- As a result, there are more than one kind of point defects. These are:
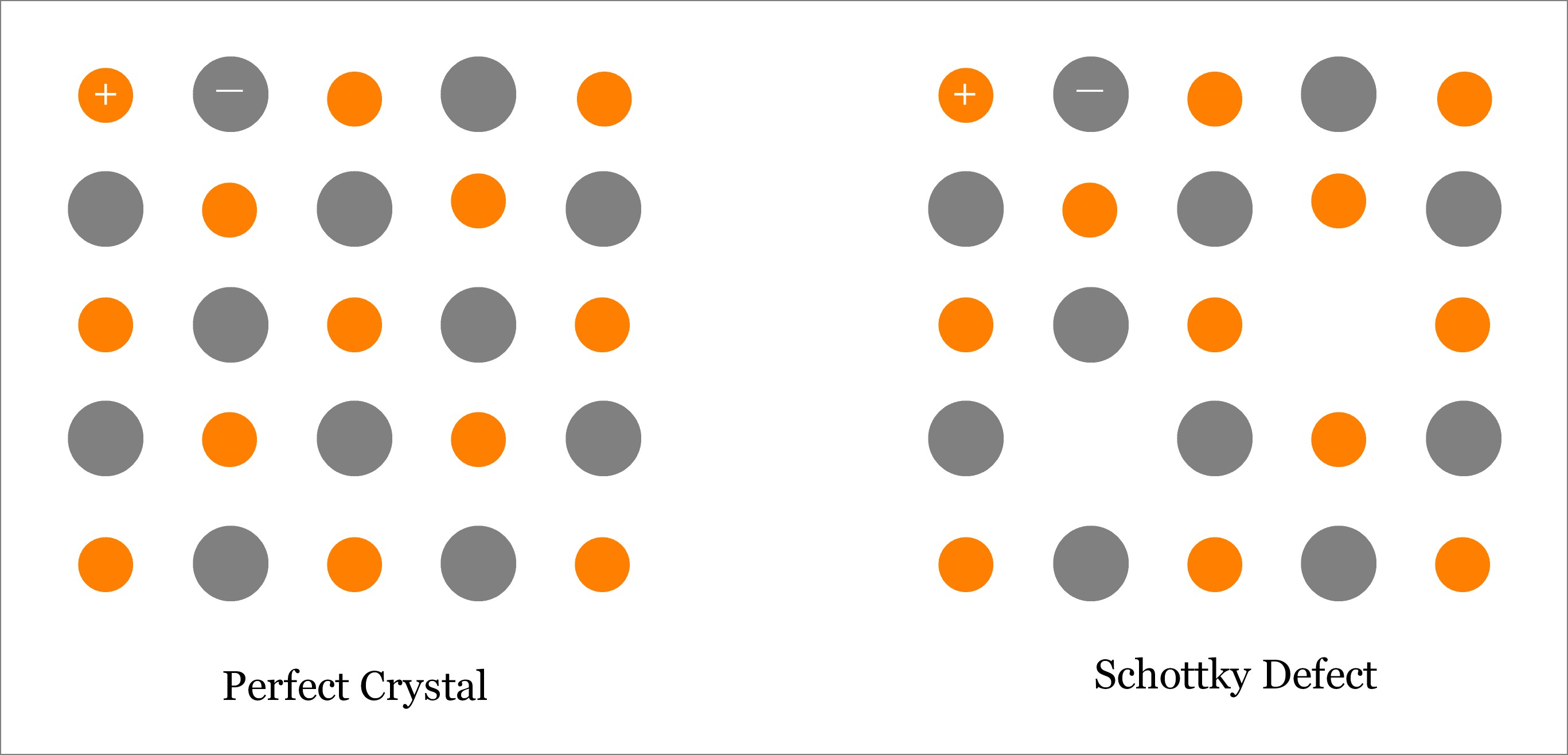
a) Schottky defect
- If an atom or ion is missing from its lattice site, the vacancy created is termed as a Schottky defect.
- In ionic solids, vacancy at a cation site is accompanied by another vacancy at a nearby anion site.
- Such paired vacancies called Schottky defects, preserve the electrical neutrality of the crystal.
- In sodium chloride, there are approximately 106 Schottky pairs per cm3 at room temperature.
- There are about 1022 ions in one cm3 of sodium chloride.
- Therefore, in sodium chloride at room temperature there is one Schottky defect per 1016 ions.
- The presence of Schottky defects in a crystal lowers its density appreciably

b) Interstitial defect
- In crystals, a part of the empty space is distributed throughout in the form of voids.
- These voids are also called interstices, or interstitial sites.
- Sometimes, certain small atoms or ions occupy these voids.
- The atoms or ions, which occupy the vacant voids in a crystal, are called interstitials.
- This defect increases the density of the crystal slightly.

c) Frenkel defect
- These are commonly found in ionic solids.
- Whenever an ion leaves its lattice site and occupies an interstitial site, such hybrid defect (combination of Schottky and interstitial) is called frenkel defect.
- In pure alkali halides, Frenkel defects are not observed, because the ions are bigger than the void size.
- Frenkel defects are found in silver halides (due to smaller size of Ag+).
- Frenkel defects do not affect the density of the crystal.
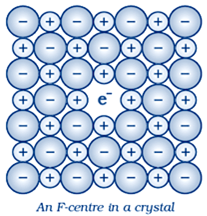
d) F- centre
- Sometimes an anion is missing from its lattice site in a crystal.
- Such single anion vacancy in an ionic crystal creates a local excess positive charge at that lattice site.
- An electron can migrate to this site and get trapped in the anion vacancy. Such a defect is called F-centre.
- If the concentration of F-centres in any crystal is high, then the otherwise colourless crystals develop some colour.
- For example, F-centres impart violet colour to KCl, and pink colour to LiCl crystals.
- In alkali halides, F-centres are created by heating alkali halide crystals in an atmosphere of alkali metal vapours.
- The metal atoms deposited on the surface of the crystal react with halide ion diffusing into the surface to give the halide.
- The electron released during this reaction diffuses into the crystal and combines with the anion the vacancy to give a F-centre.
Lattice Defects
- If the deviations from the periodic arrangement extend over microscopic regions, the defects are called lattice imperfections or lattice defects.
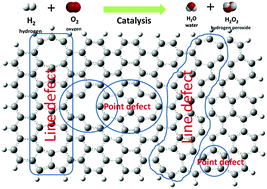

- The lattice defects are of two types. They are:
- Line defects: When a lattice defect extends along lines e.g., displacement of a row of lattice points, it is called line defect.
- Plane defects: When the irregularities extend along surfaces i.e., along the lattice plane, the defects is known as plane defect.
References:
i) https://freeguruhelpline.com/class-11-atomic-imperfection/
ii) https://physics.uwo.ca/~lgonchar/courses/p2800/Chapter4_Defects&Growth_handouts.pdf
Atomic Imperfections (Atomic Defects)

