Introduction of chemical mutagens
- It is a mutation agent which is a chemical substance that mimic nitrogen base in normal DNA without coupling during DNA replication.
- Moreover, chemical mutagen has an ability to disturb the DNA replication by inserting themselves between nitrogen bases.
- There are various types of chemical mutagens. They are as follows:
Types of Chemical Mutagens
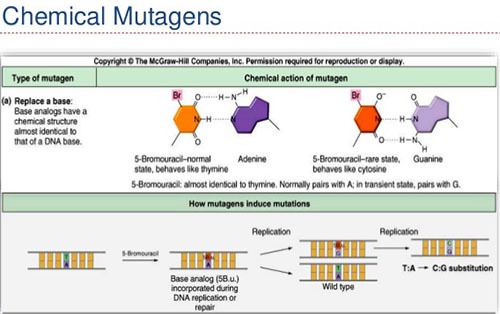
a) Base analogs
- They are those molecules that resemble purine and pyrimidine bases in structure and yet display faulty pairing properties.
- When one of these base analogs is incorporated at a site in DNA in place of natural nucleotide, replication may occur normally most of time but replications error occur at high frequencies at these sites.
- This result in incorporation of wrong base into the copied strand causing mutation.
- For examples: 5-bromo uracil (analogue of thymine), 2- aminopurine (analogue of adenine), 5-bromodeoxyuridine, 2,6-diaminopurine, etc.
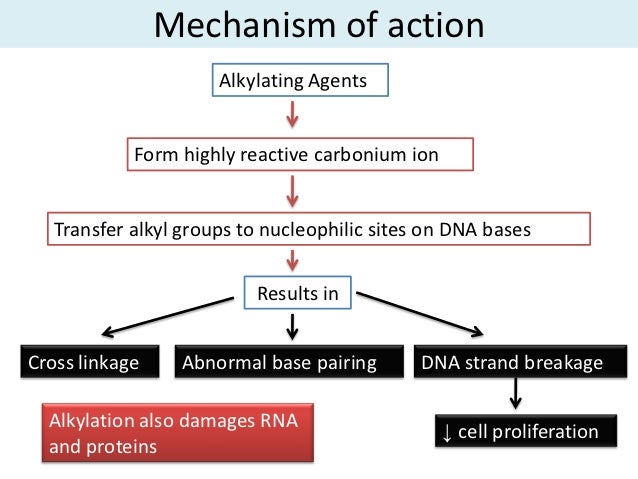
b) Alkylating agents
- They react with amino, carboxyl and hydroxyl groups in proteins and nucleic acid substituting them with alkyl groups.
- They induce mutation at higher frequencies than base analogs and are able to introduce changes even in non-replicating DNA.
- Both alkylating agents and base anlaogs tend to induce base pair substitutions.
- Alkylation increase possibility of ionization with the introduction of pairing errors.
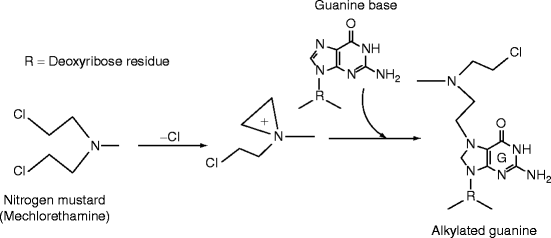
- This occurs mostly in purine (N7-guanine, N3-adenine) and alkylated base may be lost to cause de-purination.
- For examples:
Mono-functional: dimethyl sulpahte (DMS), ethyl methane sulphate (EMS) and EES.
Bi-functional: nitrogen mustards, mitomycin, nitroso guanidine, etc.
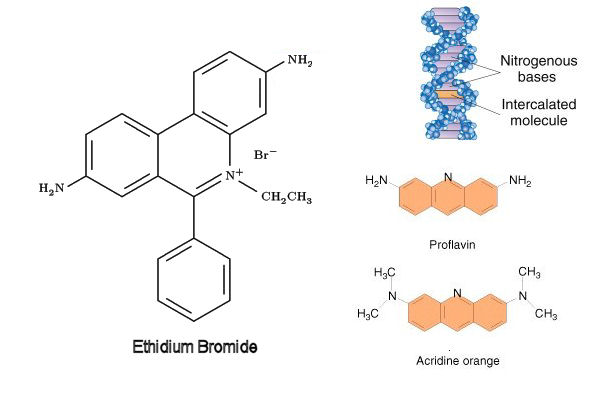
c) Inter-relative dyes
- The planar three ringed molecules having similar dimensions as those of purine and pyrimidine that can be inserted between two DNA base pairs (interaction) in aqueous solution are called intercalative agents.
- For examples: acridine orange, proflavin, acriflavin, etc.
- They typically induce frameshift mutations.
- Ethidium bromide, which is often used to detect DNA in electrophoresis, can also function as intercalative agent.

d) Deaminating agents
- Some of the amino groups in adenine, cytosine and guanine are particularly vulnerable and can be removed by many chemical agents.
- This chemical agents are called deaminating agents.
- For examples: hydroxyl amine (NH2OH) and Bisulfite deaminate only cytosine, Nitrous acid deaminates adenine, guanine and cytosine.
- Deamination of DNA bases is mutagenic because it results in base mis-pairing.
- The frequency of deamination by HNO2 is in the order A>C>G.
- Some of the examples of hydrolysis and deamination are
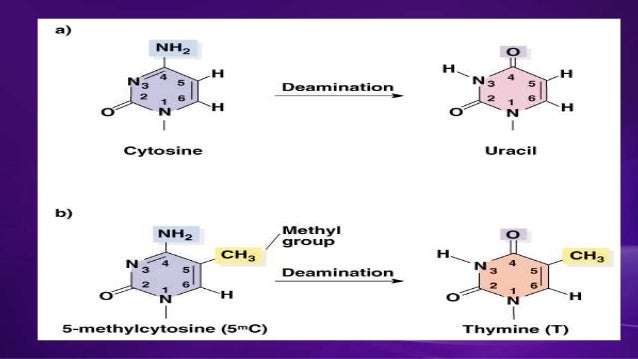
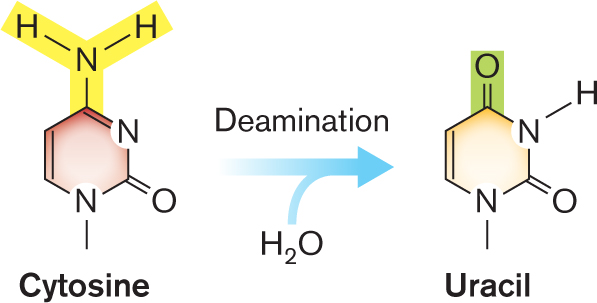
- Cytosine deaminated to uracil.
- Adenine deaminated to hypoxanthine.
- Guanine deaminated to xanthine.
- 5-methyl cytosine deaminated to thymine, etc.
e) Damage due to reactive oxygen(oxidation)
- The more reacting form of oxygen e.g. superoxide radicals, hydrogen peroxide and hydroxyl radicals are very damaging.
- They may be produced by normal cellular reactions as a result of environment factor.
- Aerobes possess superoxide dismutases, catalases and peroxide reductases to destroy reactive O2 forms.
- They have also repair enzymes and systems to repair oxidative damage to DNA.
- The accumulation of this type of DNA damage may increase the cancer rate.
- Guanine is oxidized by these reactive O2 species to 7,8 dihydro-8-oxoguanine or oxo-G.
- This oxo-G is highly mutagenic and can base pair with A and C.
- This oxidation causes G-C to T-A transition which is the most common mutation in human cancer.
- Cells have evolved many mechanisms for avoiding the resultant mutation of oxo-G.
- For example: Mut M enzyme (N-glyoxylase), Mut Y enzyme (N-glyoxylase), etc.
References:
i) https://www.biologydiscussion.com/genetics/types-of-mutagens-chemical-and-physical-genetics/65297
ii) https://onlinelibrary.wiley.com/doi/pdf/10.1111/j.1601-5223.1949.tb03333.x