- The force that acts at right angles to an imaginary line of unit length at the surface of liquid at rest is called surface tension.
- Simply, it can be defined as the force which is responsible for the tension at the surface of the liquid.
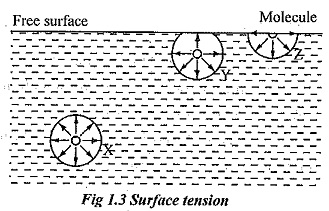
- It is one of the important properties exhibited by liquids that exist due to the intermolecular forces.
- The following important phenomena occurs due to surface tension:
A) Spherical shape of liquid drops
Surface tension accounts for the spherical shape of liquid drops.The effect of surface tension is to reduce the area of surface to minimum. Hence, liquid drops acquire spherical shape because sphere has minimum surface area for a given volume.

B) Capillary action
When one end of a capillary tube is put into liquid, the liquid rises up or falls down in the capillary tube. For example, water rises in a capillary (or rise of oils in the wick of lamp) and mercury falls in a capillary. The rise or fall of liquid in capillary is called capillary action. The surface tension of liquid is responsible for capillary action. The capillary action can be explained on the basis of the net effect of the cohesive forces and adhesive forces. The attractive forces between molecules of the same substances are called cohesive forces while that in the molecules of different substances are called adhesive forces. When the adhesive forces are greater than the cohesive forces (as in the case of water or oil), the molecules of liquid are attracted more strongly by the molecules of the glass capillary. Hence, the liquid water rises in the capillary and forms a concave meniscus. On the other hand, when cohesive forces are greater than the adhesive forces (as in the case of mercury), the liquid level inside the capillary is lower than the level outside and formed meniscus is convex.
C) Cleansing action of soaps and synthetic detergents
Cleansing action of soaps and synthetic detergents is due to their property to lower the interfacial tension between water and grease. Lowering of interfacial tension facilitates the mixing of water and grease. In other words, soap or detergent causes emulsification of grease in water and dirt can be easily washed away with water. Consequently, dirt which sticks to the grease is also removed.

Image source: wikiversity
D)Filling a water glass above the rim
The phenomenon of surface tension also explains why a glass can be filled above its rim with water. The water piles up, trying to assume a spherical shape. However, if too much water is added to the glass, the force of gravity dominates over the surface tension and the “skin” breaks and water overflows.
E) Efficiency of tooth pastes
Efficiency of tooth paste and mouth washes, in part, is based on the fact that these preparations contain substances which can lower surface tensions. Lowering of the surface tension helps these preparations to spread evenly over the surface they come in contact with. Hence, efficiency of their antiseptic action increases.
References:
i) https://chemistry-desk.blogspot.com/2011/06/important-consequences-of-surface.html
ii) https://www.quora.com/What-are-the-effects-of-surface-tension
iii) https://www.biolinscientific.com/blog/why-is-surface-tension-important

