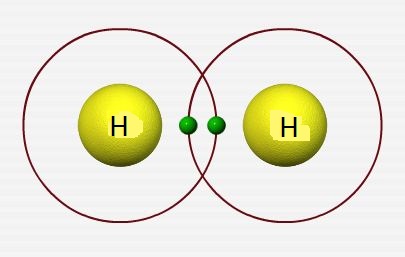
Introduction to Covalent bond
- In 1916, G.N Lewis suggested that atoms may combine with one another by mutual sharing of their valence electrons.
- Due to this, the combining atoms attain the nearest noble gas configuration in their valence cells.
- These shared pairs of electrons are situated exactly or nearly at the midpoint of the combining atoms.
- Thus, a covalent bond is the force of attraction which arises due to mutual sharing of electrons between the constituent atoms.
- The number of electrons contributed by an atom for sharing to form a covalent bond is called its co-valency.
- Covalent bond is shown by dash (-) and is also called as molecular bond.
- The single covalent bond is formed by sharing one electron each with the combining atoms, double bond is formed by sharing of two electrons each with the combining atoms and so on.

- Compounds containing covalent bond are known as covalent compounds or non-ionic compounds.
- Non-metals are electronegative elements. They have the tendency to gain electrons.
- When electronegative elements combine together they generally achieve stable noble gas configuration by mutually sharing their one or more valence electrons to form a covalent bond.
- Thus, covalent bond may be defined as the attractive force between atoms created by sharing of electron pair.
- A covalent bond is formed between two similar or dissimilar non-metallic atoms having short of one or more electrons than the nearest noble gas configuration.
Types of covalent bonds
The covalent bonds are of various types. They are as follows:
1. Non-polar covalent bonds
- When covalent bond forms between two atoms having exactly same or nearly same electro-negativity value, then the bond formed is known as non-polar covalent bond.
- Homo-nuclear molecules have non-polar covalent bonds.
- For examples: H2, O2, Br2, I2, etc.
2. Polar covalent bonds
- The bond formed between two atoms of different electronegativity values is known as polar covalent bond.
- Hetero nuclear molecules have polar covalent bond.
- For examples: HF, H2O, NH3, etc.
Properties of covalent compounds
- The properties of covalent compounds are due to covalent bonds which are listed below.
- Covalent compounds might be gases, liquids or soft solids at ordinary conditions. This is because of the weak inter-molecular forces present in between the molecules. They exist as solids only when their molecular weights are high.
- Covalent crystals tend to be hard and brittle, and incapable of appreciable bending
- The melting and boiling points of these compounds are low as compared to those of ionic crystals because the covalent bond is not so strong as ionic bond and also the atoms are less powerfully attracted towards each other.
- Most of the covalent substances do not conduct electricity because of the non-availability of free electrons or charged ions that is responsible for carrying the current.
- However, there are some substances like HCl which exhibit polarity in aqueous solutions behave like ionic substances and allow electricity to pass through them accompanied by their own decomposition thereby acting as electrolytes.
- Covalent substances are not soluble in polar solvents like water. However they are soluble in non-polar solvents like benzene, carbon di-sulphide, etc. This is because of the covalent nature of the solvent.
- However, the giant molecules are not soluble in any solvent because of the large size of the molecules.
- A very interesting property of covalent crystals is the apparent lack of sensitivity of their physical properties of their bonding type.
- These compounds give slow molecular reactions. Because it involves breaking of old bonds and then formation of new bonds which takes place in presence of sufficient energy.
- They exhibit structural and stereo isomerism.
- Shared pair of electrons are situated at the particular position in covalent bond formation. Therefore covalent bond is directional in nature.
References:
i) https://byjus.com/jee/covalent-bond/