| 1. | It is a designed apparatus where electrolysis is carried out. | It is an experimental set up or advice where chemical energy is converted into electrical energy. |
| 2. | This cell consists of a glass vessel or made up of any suitable plastic material, in which two electrodes are dipped into an electrolyte solution. | Two electrodes are dipped into the same or two different electrolyte solution in this type of cell. |
| 3. | Electric current is passed through the solution by connecting the two electrodes to a battery. | Electric current is established only when the two electrodes are joined by a conducting wire.
|
| 4. | In this cell, a chemical reaction occurs as a result of electric current. | In this cell, electrical energy is produced as a result of chemical change or chemical reactions. |
| 5. | Anode is positive electrode and cathode is negative electrode. | Anode is negative electrode here and cathode is positive electrode. |
| 6. | The redox- reaction is nonspontaneous and electrical energy is supplied to make reaction to occur. | The redox-reaction is spontaneous and is responsible for the production of electrical energy. |
| 7. | The electrons are supplied by the external battery and enter through cathode and come out through anode.
Image source: byjus |
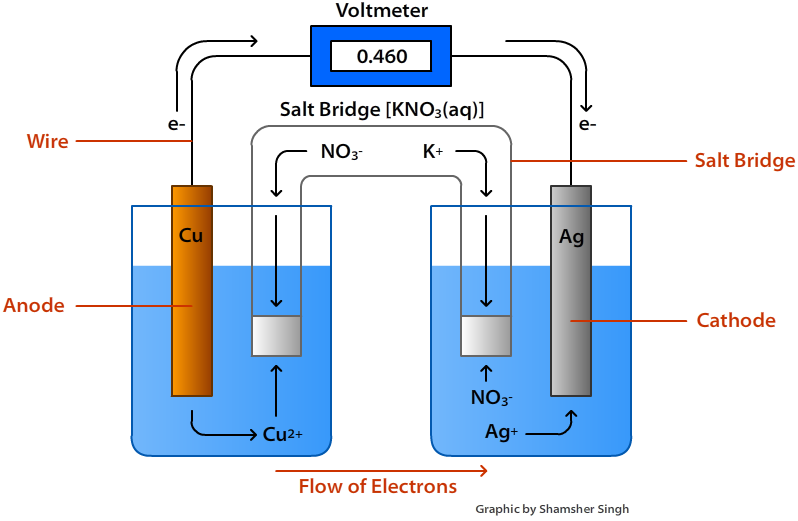
The electrons move from anode to cathode in external circuit.
Image source: chemistrylibre
|
References:
ii) https://difference.guru/difference-between-voltaic-or-galvanic-cells-and-electrolytic-cells/
Differences between electrolytic cell and voltaic cell