The differences between Salk and Sabin type vaccine are as follows:
| S.N | Salk type (IPV) | S.N | Sabin type (OPV) |
| 1. | Stands for inactivated polio vaccine. | 1. | Stands for oral polio vaccine. |
| 2. | Virus used is killed and formalized. | 2. | Virus used is live and attenuated. |
| 3. | Administered subcutaneously or intra muscularly. | 3. | Administered orally. |
| 4. | Good systemic immunity whereas mucosal slight or moderate. | 4. | Good systemic immunity whereas excellent mucosal. |
| 5. | Reversal of virulence through mutations not known. | 5. | Reversal of virulence may be possible. |
| 6. | Enteric viruses do not interfere the efficiency of vaccine. | 6. | Enteric viruses may interfere its efficiency. |
| 7. | Vaccine associated disease complication is not known. | 7. | Vaccine associated disease complication may be possible. |
| 8. | Vaccine administration is allowed to immune-compromised host. | 8. | Vaccine administration is not allowed or recommended to immune-compromised host. |
| 9. | High in manufacturing cost. | 9. | Low in manufacturing cost. |
| 10. | Jonas Salk developed this vaccine in 1954. | 10. | Albert Sabin developed this vaccine in 1961. |
| 11. | Long term immunity provided. | 11. | Longer time immunity provided than Salk.
|
| 12. | Paralysis associated with the vaccine not observed. | 12. | Paralysis associated with the vaccine is observed in very few cases.
|
| 13. | Antibody developed on the body is IgG. | 13. | Antibodies developed in the body are IgG and IgA. |
| 14. | Storage and transport does not require stringent conditions. Shelf life is longer. | 14. | Storage and transportation needs sub-zero temperatures, unless stabilized. Can be damaged easily. |
| 15. | Manufacturing is difficult. | 15. | Manufacturing is easy. |
| 16. | Controlling epidemics may not be possible. | 16. | Can be more effective in controlling epidemics. |
| 17. | Mostly used in developed countries. | 17. | Mostly used in developing countries. |
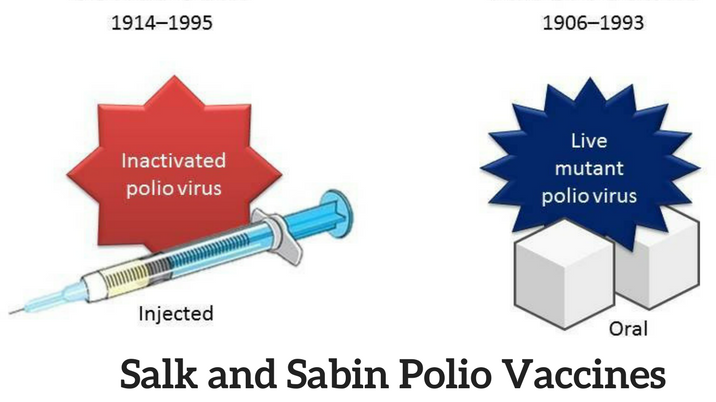
Image source: mcqbiology
References:
i) https://pubmed.ncbi.nlm.nih.gov/11763328/
ii) https://www.cdc.gov/vaccines/vpd/polio/public/index.html
iii) https://pt.slideshare.net/Muhammadiqbal583/polio-vaccine-85407875/16

