- Liquids just like solids expand on heating and contract on cooling. The expansion and contraction in liquids is more than those in solids.
- The reason for this is that the intermolecular force in liquids is less than that in solids.
- Hence, the molecules in a liquid vibrate more than the molecules in a solid when they get thermal energy.
- Since the liquids do not have a shape of their own but possess a fixed volume, we have to consider their co-efficient of cubical expansion while studying their expansion.
- When a liquid is heated in a vessel, the vessel also expands on heating. Therefore, while studying the cubical expansion of liquids, the expansion of container should also be taken into account.
- The reading of expansions of liquids would have been a little more, had there been no expansion of the vessel.
- So in case of liquids, we define two coefficients of cubical expansion.
Real and apparent expansion of a liquid
- The expansion of the liquid due to increase in temperature is always associated by expansion of containing vessel.
- The expansion which we observe is called apparent expansion which is always less than real expansion of the liquid.
- Let us take a test tube filled with coloured water. Its mouth is closed with a rubber cork having a hole. A capillary tube is inserted into the test tube through the hole in the cork as in figure.
Initially, the water rises in the capillary tube. Let the water rises up-to point A. The test tube is placed in a beaker in which water is boiled. Initially, the water level in the capillary falls. Let it falls to point B. After that, water level begins rising and it rises above the initial level. Let it rises up-to point C.
The fall in water level in the capillary in the beginning of the heating is due to the expansion of the capillary. It is because the capillary tube which is made up of glass expands more rapidly than water. So in the beginning, the capillary expands but the water does not. Therefore, we observe fall in water level.
After sometime, the water in the capillary also gets enough heat to expand. When the water starts expanding, the water level rises in the capillary. Since the expansion of the water in the capillary is more than the expansion of the capillary, the water level rises above the initial level in the capillary.
From this experiment, it is clear that a liquid like water expands on heating. This experiment shows that
- Volume of water from A to B in the capillary represents expansion of the capillary.
- Volume of water from A to C in the capillary represents apparent (or observed) expansion of water.
Hence, the real expansion of water is given by
Real expansion = expansion of the capillary + apparent expansion of water
For any liquid,
Real expansion of liquid = expansion of vessel + apparent expansion of liquid
Coefficient of real expansion (γr)
- It is defined as the real increase in volume of the liquid per unit volume per unit rise in temperature.
- That is, γr = Real increase in volume/( Original volume × rise in temperature)
Let V0 and Vθ be the volumes of a liquid at 0oC and at θoC respectively, then
γr = (Vθ – V0)/( V0 ×θ)…………………………… (i)
Vθ = V0 (1 + γr θ)…………………………………… (ii)
Coefficient of apparent expansion (γa)
- It is defined as the apparent (or observed) increase in volume per unit volume of the liquid per unit rise of temperature.
- That is, γa = Apparent increase in volume/ (Original volume × rise in temperature)
Relation between Coefficient of Real and Apparent Expansion
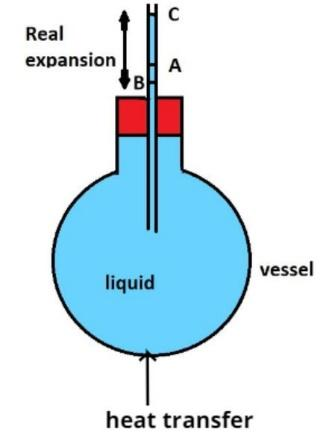
- Let us consider some liquid contained in a capillary tube at room temperature θ1oC with level of liquid at A as shown in figure. Let V be the original volume of the liquid. Suppose the system is heated to temperature θ2oC. As heat is supplied to the system, the capillary expands first and the level of liquid falls to the level B. Thus, volume ΔVg in between levels A and B gives the expansion of the capillary tube. Therefore,
ΔVg = V γg(θ2 – θ1)………………………………… (iii)
where ‘γg’ is coefficient of cubical expansion of the glass.
On heating, the liquid expands and its level moved up from B to C.
Thus, the real increase in volume ΔVr is equal to the level between B and C.
Therefore,
ΔVr = V γr(θ2 – θ1)………………………………… (iv)
where ‘γr’ is the coefficient of real expansion of liquid.
Apparently, the liquid in the capillary tube rises from A to C.
Thus, the apparent increase in volume is given by,
ΔVa = V γa(θ2 – θ1)………………………………….. (v)
where ‘γa’ is the coefficient of apparent expansion of liquid.
From the figure it is clear that,
BC = AB + AC
ΔVr = ΔVg + ΔVa
V γr(θ2 – θ1) = V γg(θ2 – θ1) + V γa(θ2 – θ1)
γr = γg + γa …………………………………………….. (vi)
Thus, the coefficient of real expansion of the liquid is equal to the sum of the coefficient of cubical expansion of the container and the coefficient of apparent expansion of the liquid.
If ‘α’ is the coefficient of linear expansion of the material of the containing vessel,
Then, γg = 3α
The equation (vi) becomes
γr = 3α + γa……………………………………………… (vii)
References:
i) https://en.wikipedia.org/wiki/Thermal_expansion

