- There are a number of steps involved in extraction of metals from its ores and is itself a science also.
- The science of extracting metals from their ores and utilizing them for various useful purposes is called metallurgy.
- The nature and properties of metals make its extraction difficult by using single method.
- Thus, it is important to know that no single process can be used for extraction of all the metals.
- The process to be used may differ from metal to metal.
- However, some steps are common that are involved in the extraction of metals. They are:
a) Crushing and Pulverization of the ore
b) Dressing or concentration of the ore
c) Extraction of the metal from concentrated ore
d) Purification or refining of the metal
A) Crushing and Pulverization
- When the ore is taken out of earth’s crust, it is in the form of big lumps.
- These are broken to smaller pieces with the help of hammers or mechanical crushers.
- Most commonly used crushers are called stone breakers or jaw crushers.
- In these crushers, there are two plates out of which one moves and the other is stationary.
- The lumps of ores are crushed between these two plates.
- The small pieces of ore are then changed into a fine powder with the help of a stamp mill or ball mill.
- The process is called pulverization of the ore.


B) Dressing or Concentration of the Ore
- Ores extracted from the earth’s crust are not pure as it contains various unwanted impurities like sand, rocky material, earthy particles, etc.
- The impurities associated with the ore are collectively called gangue or matrix.
- Before extracting the metal from the ore, it is necessary to remove the gangue.
- The process of removal of gangue from the ore is called dressing, concentration or benefaction of the ore.
- Depending upon the nature of impurities in the ore, the powdered ore can be concentrated by any of the following methods:
i) Hand picking
- If the impurities present are quite distinct from the ore, and are of large size, these may be removed by hand picking.
- This method is slow and is generally adopted in the initial stages of concentration.
ii) Gravity separation
- This method of concentrating an ore depends on the difference in specific gravities of the ore and gangue particles.
- The powdered ore is washed with an upward stream of running water.
- The lighter gangue particles are washed away while the heavier ore particles settle down and are removed from the base.
- This method is also called levigation.
- Generally oxides like tin (SnO2), hematite (Fe2O3) are concentrated by this method.
iii) Froth floatation process
- This method is based on the principle that different solids have different attraction for a liquid and are preferentially wetted by these liquids.
- Thus, certain metal sulphides such as PbS, ZnS, CuFeS2, etc. are wetted by certain oils like pine or eucalyptus oil or various organic sulphur compounds and not by only water.
- The crushed ore is vigorously agitated by a blast of air with water and a little oil such as pine oil or eucalyptus oil and also some alkali or mineral acid to facilitate the separation of gangue.
- The ore gets preferentially wetted by the oil forming the froth or scum on the surface which is skimmed off.
- Sand and other impurities are left behind in the bottom of the tank.
iv) Magnetic separation method
- This method is based on the difference in the magnetic properties of the ore and gangue.
- Ores having non-magnetic impurities or non-magnetic ores having magnetic impurities concentrated y this method.
- It is done by using magnetic separators which consists of a rubber belt moving over two rollers. One of the rollers has a magnet.
- The powdered ore is dropped on the moving belt at one end with the help of hopper.
- When the ore falls down from the belt at the other end having a magnetic roller, the magnetic impurities (or ore) are attracted by the magnet and form a separate heap from the non-magnetic ore (or impurities).
- Tin stone (SnO2) having wolframite impurities can be concentrated by this method.
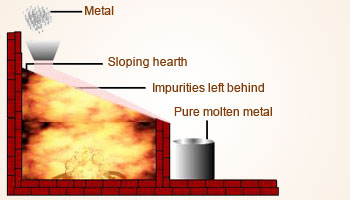
v) Liquation
- This method is used to concentrate ores having lower melting point than the impurities.
- For example, stibnite, an ore of antimony, which has low melting point, can be concentrated by this method.
- The powdered ore is heated on the slopping floor of a furnace and the temperature is adjusted to be just above the melting point of the ore.
- The ore melts and flows away while the impurities are left behind.
vi) Leaching
- This is a chemical method of concentration.
- In this method the powdered ore is treated with a suitable reagent that dissolves the ore but not the impurities.

- The impurities left un-dissolved are removed by filtration.
- Leaching method is used for concentrating ores of aluminium, silver, gold,, etc.
- For example, bauxite ore containing iron oxide as impurity is leached with 45% NaOH solution.
- Al2O3 dissolves in alkali to form soluble meta-aluminate while the impurities are left behind.
Al2O3 + 2NaOH → 2NaAlO2 + H2O
- The solution of NaAlO2 (sodium meta-aluminate) is then treated with freshly prepared Al(OH)3, when the entire aluminium in the solution gets precipitated as Al(OH)3.
NaAlO2 + 2H2O (heat) → Al(OH)3 + NaOH
- The precipitate is separated by filtration and is dried, and heated to obtain alumina.
2Al (OH)3 (heat) → Al2O3 + 3H2O
C) Extraction of the metal from the concentrated ore
- The extraction of the metal from the concentrated ore involves the following steps:
- Conversion of the ore into a compound which is suitable for reduction to the metal
- Reduction to metal
- Before the concentrated ore is subjected to final metallurgical operations, in order to get the metal in a free state, the preliminary chemical treatment may be necessary.
- The process employed for preliminary chemical treatments are calcination and roasting.
Calcination
- The process of heating the concentrated ore in a limited supply of air or in absence of air at a temperature just below its melting point is called calcination.
- It is usually done in a reverberatory furnace.
- It makes the ore porous and thus it can easily work up in subsequent stages.
- During the process of calcination, the following chemical changes occur:
- Moisture is removed.
- Volatile impurities are expelled.
- Carbonate, hydroxide and hydrated ores are converted into oxides.
Fe2O3 .2H2O (heat)→ Fe2O3 +2H2O
ZnCO3 (heat)→ ZnO + CO2
PbCO3 (heat)→ PbO + CO2
Cu (OH)2 (heat)→ CuO + H2O
Roasting
- The process of heating the concentered ore strongly in the excess supply of air below its melting point is called roasting.
- This process is generally used to convert sulphide ores into metallic oxides.
- During the process of roasting, the following chemical changes occur:
- Moisture is removed.
- Volatile impurities like arsenic and antimony are removed as their oxides.
- Organic matters are burned off and removed.
- Sulphide ores are converted into metallic oxides.
S + O2 → SO2
4As + 3O2→ 2As2O3
P4 + 5O2→ 2P2O5
2ZnS + 3O2→ 2ZnO + 2SO2
2PbS + 3O2 → 2PbO + 2SO2
2Cu2S + 3O2→ 2Cu2O + 2SO2
Smelting
- The extraction of the metals from the calcined or roasted ore consists of two steps i.e., removal of earthy impurities and reduction of the ore to metal.
- The different methods applied for carrying out the concentration of ores are useful to separate only a part of gangue and the ore still contains some earth impurities.
- There are certain substances which when mixed with furnace charge and heated, combine with the gangue to form an easily fusible material which is not soluble in molten metal.

- Such a substance which combines with gangue to form light, easily fusible material is called flux and the light, easily fusible material formed is called slag.
Flux + Impurities → Slag
(present in the ore) (Fusible product)
- Being lighter, the slag can be easily removed from the surface of the fused metal.
- Slag also prevents the oxidation of metal. The selection of flux depends upon the nature of the impurities present in the ore as given below:
a) Acidic flux: When the ore is associated with basic impurities like lime, FeO, etc., the flux used is some acidic substance like silica, borax, etc. For example:
CaO + SiO2 → CaSiO3
(Basic gangue) (Acidic flux) (Slag)
b) Basic flux: When the ore associated with acidic impurities like SiO2, P2O5, etc., the flux used is some basic structure such a CaO, CaCO3, FeO, etc. For example:
SiO2 + CaO → CaSiO3
(acidic gangue) (basic flux) (slag)
- Thus, the function of flux is to remove the non-fusible impurity as fusible slag.
- Metals are present in the ore in combination with other elements.
- From the ore, metals are obtained by the process of reduction.
- Carbon, hydrogen and metals like sodium, aluminium, magnesium are good reducing agents.
- The method of reduction and the nature of reducing agent to be used for the reduction depends upon the activity of the metals.
- Metals which are at the centers of the lower parts of the electrochemical series like iron, zinc, tin, lead, cadmium are produced by carbon reduction of their oxides at high temperature whereby the metal is obtained in the molten state.
- The process is often referred to as smelting. The process of extracting a metal by fusion of the oxide ore with a reducing agent is known as smelting.
- In this process, the roasted or calcined ore is mixed with suitable amount of coke (which acts as a reducing agent) and is heated to a high temperature above its melting point.
- During reduction, an additional substance, flux, is also added to the remove the impurities still present in the ore in the form of fusible product,
- The following methods are used for the reduction of calcined or roasted ores into the free metals.
1) Reduction with carbon:
- The carbon reduction process is also called smelting.
- The operation consists in the reduction of the oxide to the metal by strongly heating the ore with carbon in the form of coke or anthracite.
- Carbon or carbon monoxide produced by incomplete combustion of carbon, reduces the oxide to free metal. For example:
ZnO + C→ Zn + CO
PbO + C → Pb + CO
Fe2O3 + 3C → 2Fe + 3CO
Fe2O3 + 3 CO → 2Fe + 3CO2
- In order to remove the earthy impurities (gangue) in the form of fusible mass (slag) some other substance (flux) is added.
- This process is generally carried out in a blast furnace.
2) Reduction with aluminium (Gold Schmidt’s alumino-thermic process)
- When the affinity of oxygen for the metal is greater than its affinity for carbon, these metallic oxides cannot be reduced by carbon and aluminium powder is used to reduce them.
- The oxide of the metal is thoroughly mixed with aluminium powder and a little barium peroxide (BaO2) and is then placed in a crucible.
- BaO2 acts as an oxidizing agent and hence, facilitates the ignition.
- The charge is ignited with the help of magnesium wire embedded in magnesium powder and BaO2 powder.
- The reaction is highly exothermic and heat produced is sufficient to melt the metal.
- For example: Cr2O3 + 2Al → Al2O3 + 2Cr + heat
- Oxides of certain metals such as chromium, tungsten are not easily reduced by carbon monoxide.
- In such cases, aluminium which is more electropositive than chromium, manganese and tungsten, is used as reducing agent.
- The process is also known as aluminothermy or alumino-thermic process.
3) Reduction with hydrogen
- Hydrogen is very good reducing agent for reducing metal oxides. However, it is more expensive than carbon.
- The roasted ore is heated in small trays in the current of hydrogen.
- The method is suitable for metals like tungsten and indium.
- For example: WO3 + 3H2→ W + 3H2O
In2O3 + 3H2 → 2In + 3H2O
4) Reduction with water gas
- Water gas which is a mixture of CO and H2 gas can be employed for reduction in certain cases.
- Both CO and H2 gas present in the water gas act as reducing agents.
2NiO + [CO + H2] (300oC) → 2Ni + H2O + CO2
5) Self-reduction
- In certain cases, no external reducing agent is required.
- A part of the ore is oxidized to oxide or sulphate which then reacts with the rest of the sulphide ore to give the metal and sulphur dioxide.
- For example:
2Cu2S + 3O2 → 2Cu2O + 2SO2
2Cu2O + Cu2S → 6Cu + SO2
6) Electrolytic reduction (electrochemical process)
- The most reactive metals like sodium, potassium, calcium, magnesium, aluminium which occur at the top of the electrochemical series are highly electropositive and cannot be reduced by chemical reduction process.
- These metals cannot be obtained by the reduction of their oxides because the liberated metal combines with carbon at high temperature to form the carbide.
- Such metals are prepared by electrolytic reduction of their molten salts.
- The process of extraction of metals by electrolysis process is called electrometallurgy.
- For example, sodium is extracted from fused sodium chloride by electrolysis as:
NaCl → Na+ + Cl– (ionization)
At cathode: 2Na+ + 2e– → 2Na
At anode: Cl– +e– → Cl, Cl + Cl → Cl2
- Thus, sodium is liberated at cathode and Cl2 gas escapes at anode.
- Similarly, aluminium is extracted by the electrolysis of molten alumina.
7) Hydrometallurgy or metal displacement
- In this process, ore is dissolved in a suitable solvent and then the metal is precipitated by adding some electropositive metal.
- For example. In the extraction of silver from argentite, Ag2S, the ore is dissolved in sodium cyanide to give a complex salt. The solution is filtered to remove insoluble impurities. The sodium is then treated with scrap zinc when silver gets precipitated.
Ag2S + 4NaCN ⇌ 2Na[Ag(CN)2] + Na2S
2Na[Ag(CN)2] + Zn → Na2[Zn(CN)4] + 2Ag
D) Purification and refining of the metal
- The metals, thus extracted may contain impurities like residual slag or flux, unreduced oxide or sulphide of metal, substance picked from the furnaces, non-metallic elements like P, Si, S, As, etc., other metals present in the ore etc.
- The process of removal of impurities from the crude metal is termed as refining.
- Depending upon the nature of the impurities present in the metals, any of the following methods can be applied for the purification of the extracted metals.
1. Oxidation of impurities
- The method is employed for the purification of metals contaminated with impurities which are readily oxidized forming a scum on the surface of the molten metal.
- Depending upon the nature of metal, any of the following oxidation processes may be used.
i. Tossing
- The molten metal is taken on a small ladle and tossed and allowed to drop from a little height in the air.
- Impurities are thus oxidized. Lead may be oxidized by this method.
ii. Poling
- The impure metals consisting of oxide impurity can be purified by this method.
- The molten crude metal sample is stirred with poles of green wood.
- The organic matter (hydrocarbon) in the poles acts as a reducing agent and reduces the oxide impurities.
- This method is used for refining copper.
iii. Bessemerization
- The fused metal is placed in an egg-shaped furnace known as Bessemer converter.
- Compressed air is blown through the molten metals.
- Further, Bessemer converter may be lined inside with silica (Acid Bessemer Process), or with CaO or MgO (Basic Bessemer Process).
- The lining depends upon the nature of impurities.

- For example, in the pig iron purification, it is lined inside with silica when it contains Mn etc. as impurities which are oxidized to their respective oxides.
2Mn + O2 → 2MnO (basic)
MnO may further combine with SiO2
MnO + SiO2 → MnSiO3 (slag)
iv. Cupellation
- This method is applicable to those metals which contain impurities of other metals forming volatile oxides.
- The process is used to purify the silver containing lead as impurity.
- Impure metal is heated in a boat shaped bone ash shallow crucible, called cupel.
- Air is blown over the metal when lead present in the silver is oxidized to PbO (litharge) and is carried by the current of air while pure shining mass of silver is left behind.
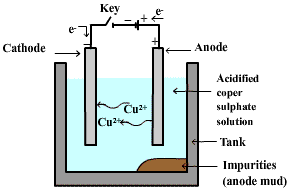
2. Electrolytic refining
- This is a very cheap and convenient method of refining metals like copper, silver, gold, aluminium, etc.
- This, most important method is based on the phenomenon of electrolysis.
- The blocks of impure metal are made the anode whereas a thin sheet of pure metal acts as cathode.
- An aqueous solution of some salt of the metal is used as an electrolyte.
- On passing current, the metal ions from the electrolyte are deposited at the cathode in the form of pure metal.
- On the other hand, an equivalent amount of the metal dissolves from the anode and passes into the electrolyte in the form of metal ions.
- The impurities either go to the solution or settle near the anode as anode mud.
3. Zone refining
- This method is particularly used when metals are required in a high degree of purity.
- Metals of very high purity can be obtained by this method by removing an impurity which shows differences in solubility in the liquid and solid states of the metal.
- A circular heater is fitted around a rod of impure metal and heater is now slowly moved along the rod.
- At the heated zone, the rod melts and as the heater passes on, pure metal crystallizes while impurities pass into the adjacent molten part.
- In this way, the impurities are swept into one end of the rod which is finally discarded.
- This process may be repeated more than once. This process is called fractional crystallization.
- This method is employed for the purification of germanium, silicon, gallium, etc. which are used in semi-conductors.
4. Van Arkel method
- This method is employed to obtain ultra-pure metals such as zinconium and titanium.
- The impure metal is converted into volatile unstable compound (generally metallic oxide) which is then decomposed by heating, resulting, in the formation of pure metal.
- For example:
Ti + 2I2 (2500C) → TiI4 → Ti + 2I2
5. Distillation
- This method is used for metals like zinc, cadmium, and mercury which make vapours easily.
- In this method, the impure metal is heated in a vessel. The metal forms vapours which are condensed separately in a receiver.
- The non-volatile impurities are left behind.
References:
i) https://edu.rsc.org/cpd/the-extraction-of-metals/4010857.article


