- The ionization energy depends upon the following factors:
A) Size of the atom
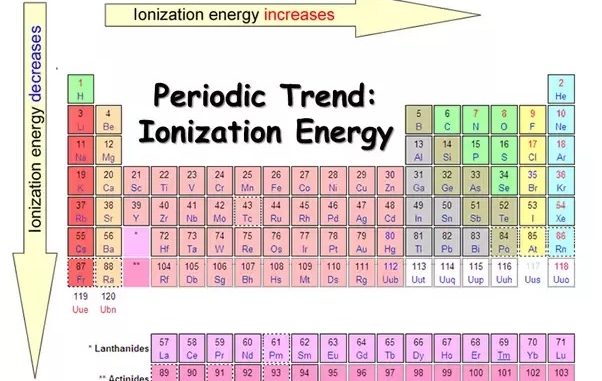
- The ionization energy depends on the distance between the electron and the nucleus i.e., size of the atom.
- The attractive force between the electron and the nucleus is inversely proportional to the distance between them.
- Consequently, as the size of the atom increases, the outermost electrons are less tightly held by the nucleus.
- As a result, it becomes easier to remove the electron.
- Therefore, ionization energy decreases with increase in atomic size.
B) Magnitude of nuclear charge
- The attractive force between the nucleus and the electrons increases with the increase in nuclear charge provided their main energy shell remains the same.
- This is because the force of attraction is directly proportional to the product of charges on the nucleus and that on the electrons.
- Therefore, with the increase in nuclear charge which becomes more difficult to remove an electron and ionization energy increases.
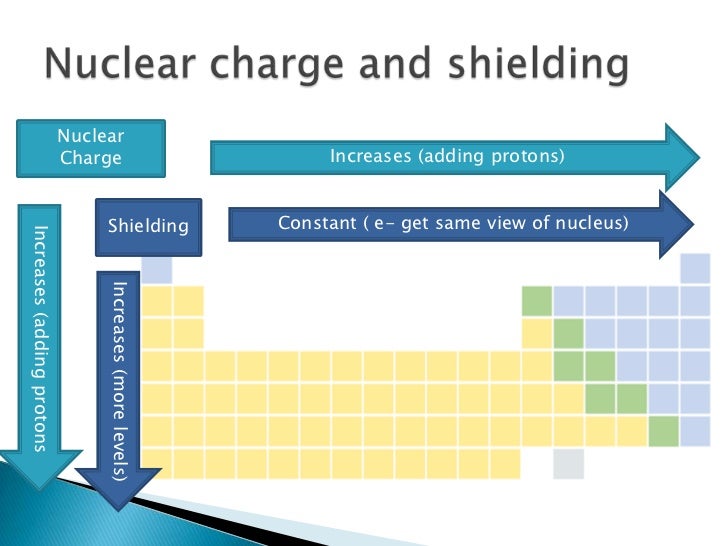
C) Screening effect or shielding effect
- In multi-electron atoms, the electrons present in the outermost shell do not experience the complete nuclear attraction because of repulsive interactions of the in between electrons.
- Thus, outer most electrons are shielded or screened from the nucleus by the inner electrons.
- This is known as shielding or screening effect.
- As a result of this, the actual charge felt by the electron is termed as effective nuclear charge.
- Effective nuclear charge (Zeff) is given by
Zeff = Total nuclear charge (Z) – Screening constant (s).
Where, screening constant takes into account the screening effect of the inner electrons.
- If the number of electrons in the inner shells is large, the screening effect will be large.
- As a result, the attractive interaction between the nucleus and the outermost electrons will be less.
- Consequently, ionization energy will decrease.
- Thus, if other factors do not change an increase in the number of inner electrons, it tends to decrease the ionization energy.
D) Penetration effect of the electrons
- It is well known that in case of multi-electron atoms, the electrons in the s-orbital have the maximum probability of being found near the nucleus and this probability goes on decreasing in case of p, d and f-orbitals.
- In other words, s-electrons are more penetrating towards the nucleus than p-electrons.
- The penetration power decreases in a given shell (same value of n) in the order
s > p > d > f.
- Now, if the penetration of the electron is more, it will be closer to the nucleus and will be held firmly.
- Consequently, ionization energy will be high.
- This means the ionization energy increases with increase in penetration power of the electrons.
- Thus, for the same shell, it is easier to move the p-electrons in comparison to s-electrons.
E) Electronic configuration
- It has been noticed that certain electronic configurations are more stable than the others.
- The atom having a more stable configuration has less tendency to lose the electron and consequently has high value of ionization energy.
- For example,
a) The noble gases have stable configuration (ns2np6). They have highest ionization energies within their respective periods.
b) The elements like N (1s2, 2s2, 2px1, 2py1, 2pz1) and P (1s2, 2s22p6, 3s2, 3px1, 3py1, 3pz1) have configurations in which orbitals belonging to same subshell are exactly half filled.
Such configurations are quite stable and consequently require more energy for the removal of electron. Hence their ionization energies are relatively high.
c) The elements like Be (1s2, 2s2) and Mg (1s2, 2s22p6, 3s2) have all the electrons paired. Such configurations being stable also result in the higher values of ionization energies.
References:
i) https://www.learningtopnotch.com/2020/05/factors-affecting-ionization-energy-and.html
ii) https://byjus.com/chemistry/periodic-trends-in-ionisation-enthalpy-of-elements/

