- The constituent particles (atoms or molecules) of a crystal are closely packed and held together by strong mutual force of attraction.
- This force which exists between different atoms of a crystalline solid is called binding forces.
- The property of a solid depends upon the nature of the binding forces. There are four types of binding forces. They are:
- Ionic binding
- Covalent binding
- Metallic binding
- Van der Waals binding
A) Ionic Binding
- An ionic crystal is made up of positive and negative ions placed in alternate positions.
- Since the magnitudes of the positive and negative charges on the ions are equal, hence, the crystal as a whole is neutral.
- In such a crystal, electrostatic forces of attraction hold the cations and anions together.
- In general, the formation of an ionic crystal can be shown as in the figure where a positive ion A+ combines with the negative ion B– to give a molecule AB.
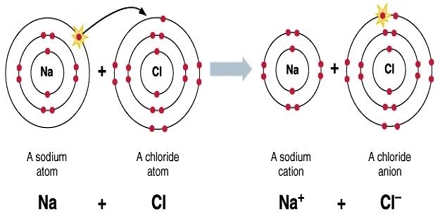
- When two such ion pairs come closer, the force of attraction between the ions of one pair and the oppositely charged ions of the other pair is set up.
- Inorganic compounds like NaCl, Na2SO4 and KOH are examples of ionic crystals.
Properties of ionic binding
i) The ionic crystals are hard and brittle.
ii) They are poor conductors of heat and electricity.
iii) They have high melting and boiling points.
iv) They are readily soluble in solvents like water (H2O) and liquid ammonia (NH3).
B) Covalent Binding
- When two similar or dissimilar atoms achieve stability by sharing valence electrons between themselves then covalent types of crystal is formed.
- Because of the sharing of the electron, the covalent binding arises.
- The binding between hydrogen (H2), chlorine (Cl2), methane (CH4), silicon, quartz, diamond etc., are the examples of covalent bindings.

Properties of covalent binding
i) These bonds are very strong which results in the covalent crystal becoming very hard.
ii) They are poor conductors of heat and electricity.
iii) They are insoluble in water but soluble in non-polar solvents such as benzene and carbon tetrachloride.
iv) They have high melting and boiling point. For example, diamond melts at around 3770 K.
C) Metallic Binding
- In metals, each atom loses all its valence electrons and becomes a positively charged ion.
- These free electrons form a kind of sea or cloud of electrons, which can move throughout the volume of the metal.
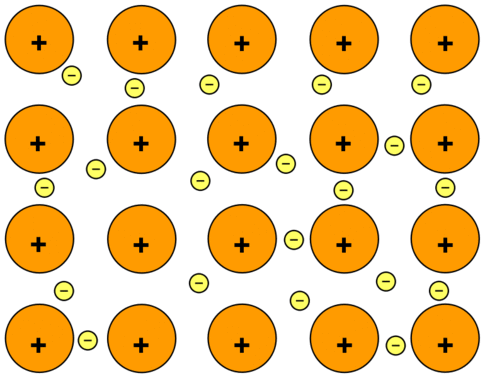
- Thus, a metal can be considered as the collection of positively charged ions immersed in a sea or cloud of electrons.
- Therefore, valence electrons are not bound directly to a particular atom but they move freely in the sphere of influence of other atoms and are bound to different atoms at different times.
- In metallic binding, the bonding is provided by the electrostatic forces of attraction between the electrons and the positive ions.
- In order to keep these metal ions in position, the electrostatic forces must overcome the mutual forces of repulsion.
- Thus, free electrons cannot escape from the body of the metal.
- The binding that exists in metals like Cu, Al, Fe, etc. are examples of metallic binding.
- The metallic binding is comparatively weaker than the ionic and covalent binding.
Properties of metallic binding
i) They have high electrical and thermal conductivity due to the availability of unlimited number of free electrons.
ii) They are malleable and ductile.
iii) They have usually moderate to high melting points.
iv) They are opaque to light. This is because the free electron in a metal absorbs light energy.
v) They possess metallic lusture.
D) Van der Waals binding
- In the inert gases like helium, neon and argon, the outermost shell is completely filled with electrons (i.e., their duplet or octet state is completed).
- In such a stable condition, the binding like ionic, covalent and metallic cannot take place because they all require the adjustment of valence electrons.
- However, atoms of all inert gases have a very weak force of attraction between each other.
- These attractive forces of attraction are called Van der Waals forces of attraction.
- These are weak binding forces. Due to this force, inert gases can be condensed into solids at very low temperature.

Properties of Van der Waals binding
i) These crystals have low densities as they are formed of elements with low atomic number.
ii) These crystals are good insulators because in such a solid, no valence electrons are available.
iii) These crystals have low melting points as Van der Waals forces results in weak molecular bond.
iv) These crystals are usually transparent to light.
v) They are soluble in both polar and non-polar liquids.
References:
i) https://physics.ucf.edu/~lc/Unit-4.pdf
ii) https://sciencing.com/types-bonding-crystals-6891243.html
iii) https://courses.physics.illinois.edu/phys460/fa2006/slides/460-lec6.pdf

