- All spontaneous processes tend to achieve a state of minimum energy and maximum entropy.
- For a system, generally it is not possible to achieve both, the minimum energy and maximum entropy simultaneously.
- There are cases where either of the two factors is favourable.
- For example, many endothermic reactions are spontaneous because the favourable entropy factor (ΔS is highly positive) outweighs the unfavourable entropy factor (ΔH is positive).
- On the other hand in some reactions the unfavourable entropy factor (ΔSsys < 0 i.e., ΔSsys is negative) is outweighed by the favourable enthalpy factor (ΔH is negative).
- From these observations, it becomes clear that ΔH (or, ΔE), nor ΔSsys alone can predict whether a reaction is spontaneous or not.
- However, the quantity, ΔStotal = ΔSsys + ΔSsurr can be used in predicting the spontaneity of a process.
- For a spontaneous process, ΔStotal should be positive.
- For chemical systems, however, it is not always possible to determine ΔSsurr.
- Under these conditions, it was felt that a new thermodynamic function is needed to predict the direction of spontaneity.
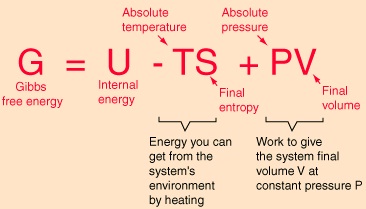
Image source: Weebly
- Gibbs’ free energy function was introduced by J. Willard Gibbs to predict the direction of spontaneity.
- This function is denoted by the symbol G.
- The Gibbs’ free energy function (G) accounts for the enthalpy and entropy changes in a system and is defined by
G = H – TS…………………………………..(i)
Where, H is the enthalpy of the system; S is the entropy of the system; T is the temperature of the system in Kelvin scale.
Since, H, T, S are state functions, hence, G is also a state function.
- The Gibbs’ free energy is defined as the amount of energy available for doing useful work under constant temperature and pressure conditions.
- The free energy function (G) as such is not very useful. Instead, the change in free energy function ΔG is very useful in predicting the state of any system.
- The ΔG is generally termed as the free energy change.

Image source: Researchgate
Suppose a system is in its initial state, say state 1.
Let it be changed to another state, say state 2, under constant temperature and pressure conditions. Then, we can write,
State 1: G1 = H1 – TS1 State 2: G2 = H2 – TS2
The change in free energy ΔG is then given by
ΔG = G2 – G1 = (H2 – TS2) – (H1 – TS1) = (H2 – H1) – T (S2 – S1)
Or, ΔG = ΔH – T ΔS
This equation is known as Gibbs’ – Helmholtz equation.
Gibbs’ Free Energy Change and Spontaneity of a Process
For a system which is not isolated, the direction of spontaneity can be predicted by the total energy entropy (ΔStotal). ΔStotal is given by,
ΔStotal = ΔSsys + ΔSsurr………………………………..(ii)
If a reaction is carried out at constant temperature and pressure, and heat equal to ΔH is given out to the surrounding, then,
ΔSsurr = qrev/T = – ΔHsys/T…………………………….(iii)
Substituting equation (iii) in equation (ii), we get,
ΔStotal = ΔSsys – (ΔHsys/T)
Since, all the terms on the right refer to the system, hence the subscript sys is dropped. The above equation than can be written as
T ΔStotal = TΔS – ΔH
Or, -T ΔStotal = ΔH – TΔS………………………………..(iv)
We know that,
G = H – TS
Or, ΔG = ΔH – TΔS – SΔT
For a change conducted at constant temperature and pressure, since ΔT = 0, hence
ΔG = ΔH – TΔS…………………………………………….(v)
Comparing equation (v) and (iv) one gets,
ΔG = – TΔStotal…………………………………………..(vi)
Thus, from above equation we can say that ΔG can also be used for predicting the direction of spontaneity.
References:
i) https://goldbook.iupac.org/terms/index/A
ii) https://link.springer.com/article/10.1557/mrs.2019.162

