- The process of mixing of the atomic orbitals is called hybridization.
- There are different types of hybridization like sp2, sp3, sp, sp3d, sp3d2, sp3d, dsp2,d2sp3.
- The atoms of third period elements have vacant d orbitals.
- Since, the energy of 3d orbitals is comparable to the energy of the occupied 3s and 3p orbitals (the energy difference is very small), hence quite often d orbitals also get involved in the bonding process.
- The paired electrons in the 3s or 3p orbitals can be promoted to the vacant 3d orbitals, thereby, opening up the possibilities of hybridization of s, p and d.
- the different types of hybridization involving only s, p and d orbitals are:
i) The sp3d hybridization: Trigonal bi-pyramidal hybridization
- In sp3d hybridization, one s, three p and one d orbital (dz2) of the same energy level (e.g., n=3) are involved in hybridization.
- These five orbitals combine to give five hybrid orbitals.
- These hybrid orbitals designated as sp3d orbitals, are oriented towards the corners of trigonal bi-pyramid.
- Three orbitals forming a plane are directed towards the corner of the equilateral triangle (angle of inclination = 1200), while the other two are oriented at right angles to the plane of the triangle, above and below it.
- A typical example of this type of hybridization is the formation of PCl5 molecule.
- The ground state outer electronic configuration of phosphorous is 3s2 3px1 3py1 3pz1 3d0.
- The promotion of one 3s electron to 3d level gives the outer configuration, 3s1 3px1 3py1 3pz1 3d1.
- These five orbitals hybridize to give five new sp3d orbitals.
- Each hybrid orbital is singly occupied.
- One of the p orbitals of chlorine is singly occupied.
- These singly occupied p orbitals of Cl form five P-Cl bonds due to the overlap with five sp3d orbitals.
- This gives a trigonal by-pyramidal geometry for PCl5 molecule.
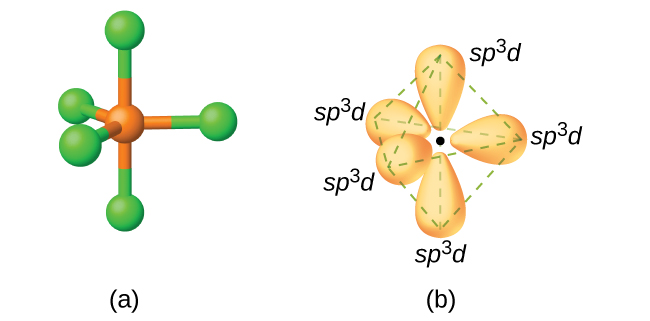
Image Source: Nigerianscholars
ii) The sp3d2 hybridization : Octahedral hybridization
- The orbitals which take part in sp3d2 hybridization are ns, n(px, py, pz), n(dz2, dx2-y2). E.g., 3s, 3(px, py, pz), 3(dz2, dx2-y2).
- In this case, the total number of atomic orbitals which hybridize is six.
- The six hybridized orbitals are directed towards the corner of an octahedron, and are inclined at an angel of 900 to each other.
- The sp3d2 hybridization is observed for sulphur in SF6.

- The ground state outer electronic configuration of sulphur is 3s2 3px2 3py1 3pz1 3d0.
- Promotion of two electrons, one each from 3s and 3px to two 3d (dz2, dx2-y2) orbitals, followed by hybridization yields six sp3d2 hybrid orbitals.
- These six hybrid orbitals are directed towards the corner of a regular octahedron.
- Each hybrid orbital is occupied by one electron.
- These hybrid orbitals overlap with the half-filled p orbitals of F atoms to form six S-F bonds. This gives an octahedral geometry for SF6.
iii) The d2sp3 hybridization: Octahedral hybridization
- The d2sp3 hybridization involves two d orbitals belonging to (n-1)th energy level and the s and p orbitals belonging to nth energy level, i.e., (n-1) (dz2, dx2-y2), ns, n(px, py, pz).
- For example: 3dz2, 3dx2-y2, 4s, 4(px, py, pz) orbitals.
- A typical example of this type of hybridization is the formation of a complex ion, [Co(NH3)6]3+.
- The outer configuration of Co is 3s2 3p6 3d7 4s2.
- Therefore, the configuration of Co3+ is 3s2 3p6 3d6 4s0 4p0.
- When the reaction takes place, the six electrons in 3d orbitals pair up. This makes two d-orbitals available.
- These two 3d orbitals combine with one 4s and three 4p orbitals to form six d2sp3 hybrid orbitals.
- These six orbitals are oriented along the six corners of a regular octahedron.
iv) The dsp2 hybridization : Square planar hybridization
- In this type of hybridization, one d orbital belonging to (n-1)th energy level, and one s and two p orbitals belonging to the nth energy level, viz., (n-1) dx2-y2, ns, npx, npy, npz.
- For example: 3 dx2-y2, 4s, 4px, 4py, 4pz are involved.
- A typical example of this kind of hybridization is the formation of complex ion [Ni(CN)4]2-.
- Nickel (atomic no. = 28) has the outer configuration 3s2, 3p6, 4s2, 3d8. The electronic configuration of Ni2+ is 3s2 3p6 3d8.
- During the reaction, these eight d electrons pair up to occupy four of the d orbitals. This leaves one of the 3d orbitals vacant.
- This 3d orbital combines with one 4s and two 4p orbitals to give four dsp2 hybrid orbitals.
- Thee four hybrid orbitals are directed towards the four corners of a square in the xy plane.
- This gives a square planar geometry to the complex ion [Ni(CN)4]2-.
References:
i) https://www.masterorganicchemistry.com/2018/01/16/a-hybridization-shortcut/
ii) https://www2.chem.wisc.edu/content/hybridization#
iii) https://www.organicchemistrytutor.com/topic/hybridization/

