- Immunoglobulins are specialized serum proteins which are produced in response to an antigen and react with that antigen specifically.
- It may also react with the very closely related antigen as well.
- They are found in various secretions of body and some are fixed to body cells or tissues in addition to humoral antibodies.
- They are generally glycoproteins and comprise 20 to25 % of the total serum proteins.
- Serum globulins are of two types. One is water soluble pseudoglobulin and the other water insoluble
- Most of the antibodies are euglobulins.
- Immunoglobulins denote the chemical structure of protein where antibody denotes biological activity and function of protein.
- They include antibody globulins, abnormal plasma proteins of myeloma, cryglobulinemia, macroglobulinaemia and naturally occurring subunits of immunoglobulin.
- Thus, all immunoglobulins may not be antibodies.
- Plasma cells principally synthesize the immunoglobulins and to a certain extent by B lymphocytes as well.
- Immunoglobulins have 5 main classes: IgG, IgA, IgM, IgD and IgE.
Structure of IgG
- It is the major immunoglobulin component of serum which makes up about 85% of the total amount (12mg/ml blood).
- It has a molecular weight of 150000 daltons in man.
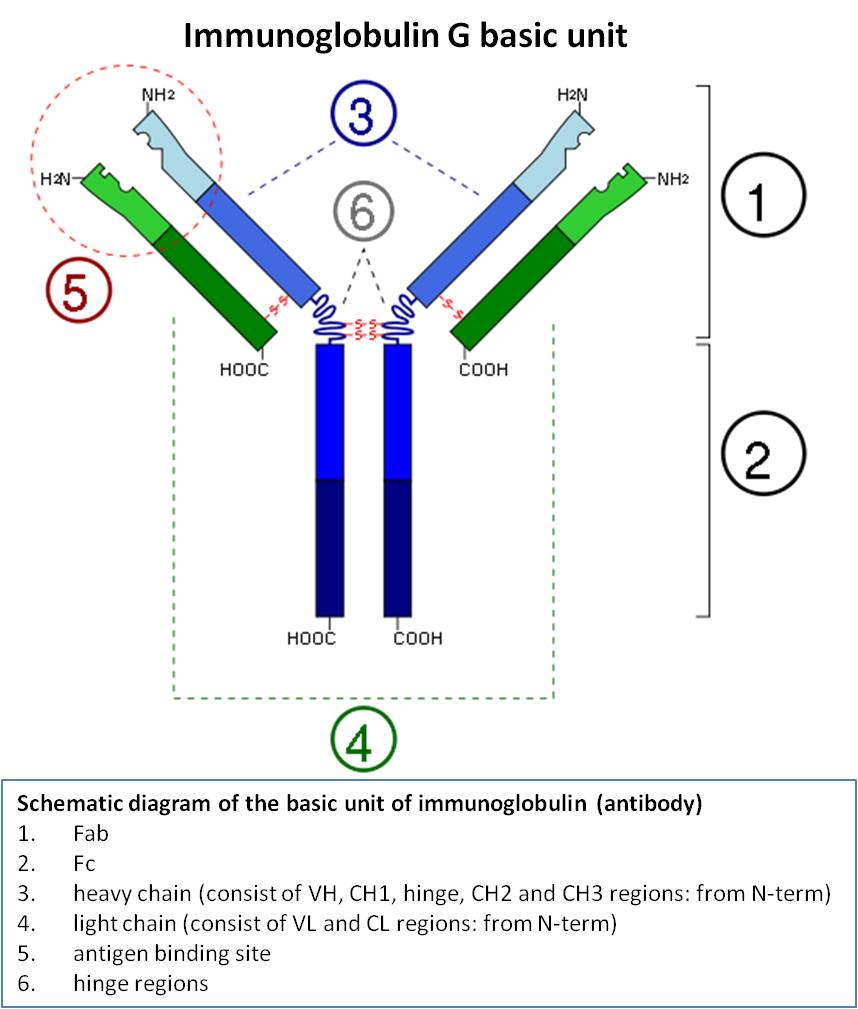
- The molecule has two antibody combining sites which is known as Fab (antigen binding) portions having parts of both the heavy and light chains.
- It is called divalent as it has got two identical antigen binding sites.
- It consists of 2H chains of 55000 daltons each linked by disulphide bonds and also of 2L chains of each 22000 daltons.
- The light chains consist of two parts which are joined together, one with a constant sequence of amino acids at the C terminal of a chain that are common to the light chains of the species under study.
- The other part at the N terminal in which variation occurs in the sequence of the 107 amino acids differing between one myeloma and another, being similar only within a particular myeloma.
- The amino acid sequences have been worked out in various species by the analysis of myeloma proteins.
- The positions in the N terminal portion have been found to be variable around 50%.
- The variable portions of light and heavy chains are contained in the Fab portion of the molecule.
- It is the portion where antibody combining site is present.
- The light chains are of two distinct types known as K or L chains.
- A single gene is responsible in controlling the entire range of variability in the light chains as no species specific amino acid residues have so far been found in the variable portion.
- Both K and L chains are produced in any individual but they are not found in the same immunoglobulin molecule.
- K and L chains are present in the ratio of about 2:1 in any one individual.
- Two intra-chain disulphide bonds occur in almost exactly the same position in both K and L chains.
- Both of these chains have cysteine as the terminal amino acid at the carboxyl end.
- This provides or serves the attachment of light chains to the heavy chains.
- These facts may give us idea about the common origin of both the chains which might have been diverged during evolution and have retained many common structural characteristics.
- The heavy chains are however specific for each class.
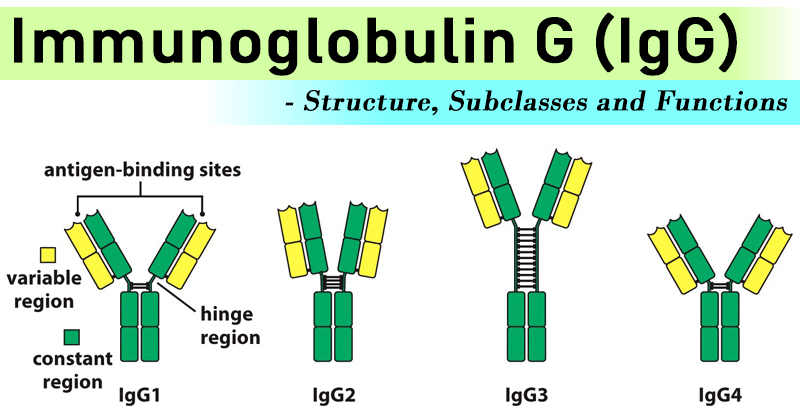
- The heavy chain (gamma chain) of IgG molecule exists in four different forms in the human.
- They are IgG1, IgG2, IgG3 and IgG4 and can be identified with specific antisera that are able to detect differences in the Fc fragment of the heavy chains.
- Among them, IgG1 occupies 65%, IgG2 -23%, IgG3 -8% and IgG4 -4%.
Imagse source: microbenotes
- IgG2 production is responsible in immunological response against the bacterial polysaccharide.
- IgG is one of the major activators of classical complement pathway, particularly IgG1 and IgG3.
- IgG has three cellular receptors- FcrR1, FcrR2 and FcrR3 which bind, recruit and activate cells such as polymorphs, macrophages and NK cells.
- FcR receptor is also responsible for active process of placental transfer from mother to foetus.
Properties and Functions of IgG
- It is a simple monomer made up of basic 4 polypeptide chains.
- It requires the help of T cell to get produced.
- It generally appears later i.e. around 2 weeks after infection but persists for longer duration.
- It is the predominant antibody if we talk about the secondary immune response.
- The sedimentation coefficient of IgG is 7S.
- There is participation of IgG in most of the immunological reactions such as precipitation, complement fixation and neutralization of toxin and viruses.
- The diffusion rate of IgG is more as compared to other immunoglobulins into extravascular spaces for which it is equally distributed in between intravascular and extravascular compartments.
- There is no formation of IgG in foetus in significant amount.
- Among all the immunoglobulins, it has got the longest half-life which is about 23 days.
- This immunoglobulin can only pass the placenta and provides natural passive immunity to the newborn.
- The total IgG concentration of serum determines the catabolic rate making its catabolism unique.
- IgG level increases in myeloma or kalaazar where the synthesized IgG against a particular antigen is catabolized rapidly.

Image source: mepedia
- This may result in the deficiency of the particular antibody.
- Conversely, IgG antibody is slowly metabolized when administered in hypogammaglobulinaemia for therapeutic purposes.
- The homologous antibody synthesis gets suppressed by feed-back mechanism when it is passively transferred to an individual.
- This principle is utilized in isoimmunization of women by anti-RhD IgG immunoglobulin during delivery.
References:
i) https://microbenotes.com/immunoglobulin-g-igg-structure-subclasses-and-functions/
ii) https://me-pedia.org/wiki/Immunoglobulin_G