- Colloidal solutions can also be classified on the basis of constitution of the particles of the dispersed phase.
- The tree types of colloids are given as below:
A) Multi-molecular Colloids
- This is the type of colloid in which the colloidal particles consists of aggregates of atoms or small molecules, having diameters of less than 1nm.
- These atoms and molecules are held together by van der Waals’ force.
- For example, gold sol may contain particles of various sizes having several atoms of gold.
- Similarly, sulphur sol consists of particles containing about a thousand of molecules of S8 .
B) Macromolecular Colloids
- Certain substances form large molecules having the dimensions of colloid particles. Such molecules are called macro molecules.
- Macromolecules have very high molecular masses.
- Since, macromolecules have dimensions comparable to those of colloidal particles hence their dispersions in any suitable medium behave like a colloidal solution.
- Such colloidal solutions are termed as macromolecular colloids. Most lyophilic sols are macromolecular colloids.
- Gelatin, starch, nucleic acids, cellulose etc. are naturally occurring macromolecules.
- Some are prepared by polymerization of simple molecules, e.g., polythene, poly-propene, polyvinyl alcohol, bakelite, etc.
Characteristics
- Some important characteristics of macromolecular colloids are given below:
- Some of the macromolecules dissolve in water and give homogeneous solutions.
- Solutions of macromolecules behave like lyophilic colloids.
- There is considerable interaction between the solute and the solvent in macromolecular sols.
- Due to the large size and shape of the macromolecules, the macromolecular colloids cause serious deviations from the solutions obeying ordinary laws.
- The solutions of macromolecular colloids have high viscosities even at very low concentrations of the solute. The high increase of viscosity may be due to the higher degree of solvation of the macromolecules. It may be caused by the immobilization of solvent molecules between the moving macromolecules. Such immobilization of water is found in aqueous solutions of gelatin, cellulose, etc.
- Macromolecules may be precipitated from their sols by adding a liquid in which the solute is insoluble. In such precipitations, the molecules with higher molecular mass separate out first, followed by the separation of the lighter molecules. Thus, by this method, the macromolecules may be separated into several fractions of different molecular masses.

C) Association (or Associated Colloids)
- There are certain substances which behave as normal strong electrolytes at low concentrations, but behave like colloid at high concentrations.
- This occurs due to the formation of the aggregated particles at higher concentrations.
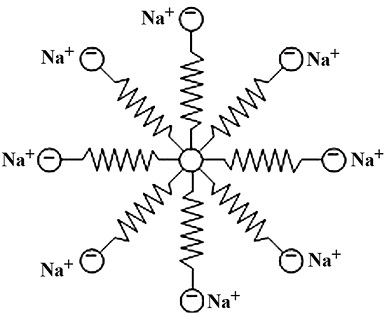
- Such substances are called association (or associated) colloids. The aggregated particles are called micelles.
- These colloids are formed by the association of long chain organic compounds such as fatty acids, quarternary ammonium compounds, etc.
- The surface of the associated ion carry the ionic charge and the counter ions in the solutions get oriented near the surface.
- As a result of which an electrical double layer is produced. This double layer is responsible for the colloidal behavior of such associated ions.
- Soap is a typical example of association colloids.
- Measurements of the colligative properties of concentrated soap solutions indicated that the solute is in the associated form.
- Soaps in aqueous solution forms ionic aggregates called ionic micelles.
- An ionic micelle contains aggregate of 50-100 ions with a considerable amount of water molecules.
- The micelles are nearly spherical in shape and are believed to be formed as follows.
- Soaps are alkali metal salts of long chain carboxylic acids, viz., RCOONa, where R is a long chain alkyl group.
- In aqueous solutions, soap ionizes as follows:
RCOONa (soap) → RCOO–(carboxylate ion) + Na+
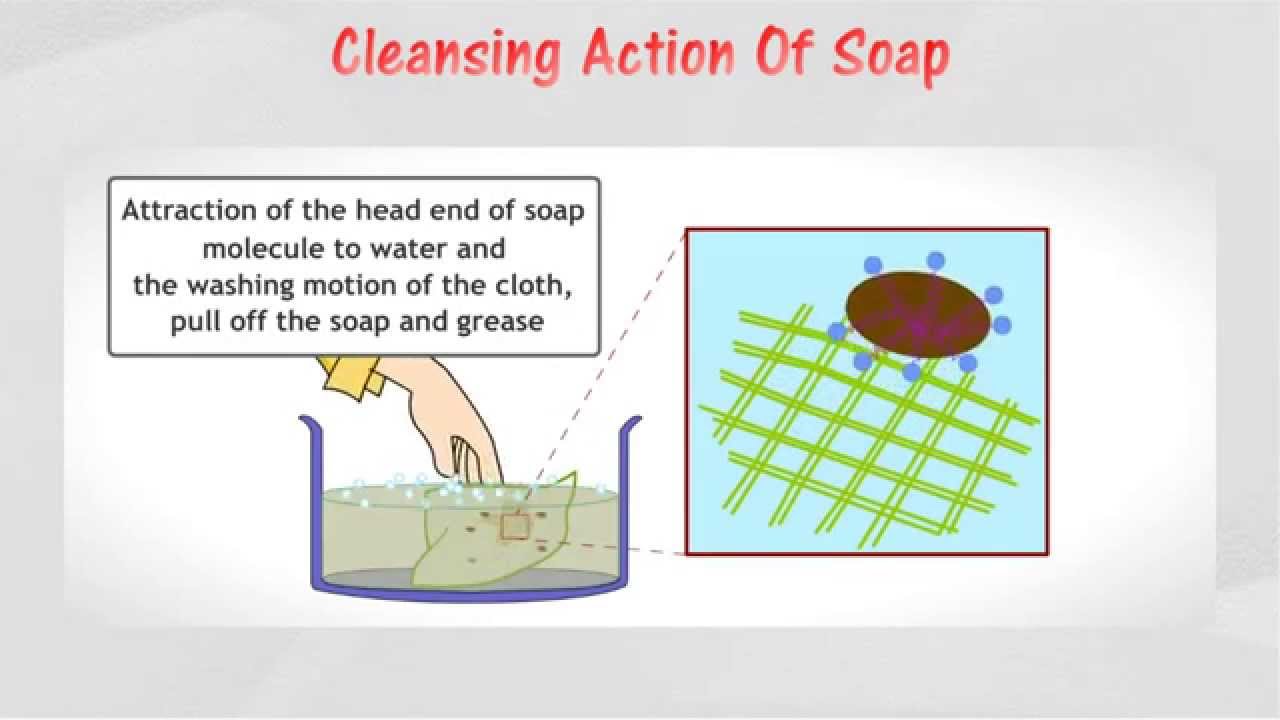
- Most common soap is sodium stearate, C17H35 The hydrocarbon residue R in RCOONa (soap) is water repellant, whereas the-COO– group is water attracting.
- So, RCOO– ion orients itself in such a way that –COO– end dips into the water, and the R group stays away from the water.
- When such RCOO– groups orient themselves on water surface, COO– groups tend to stay away from each other due to electrostatic repulsion.
- The R-groups tend to form bunch in the interior.
- The outer surface consisting of –COO– groups thus remains in contact with water, and stabilizes the micelle in water.
- So, all soap micelles are negatively charged colloidal particles. The negative charge on the colloidal particle is balances by the positive Na+ ions in the solutions.
References:
Multi-molecular, Macromolecular and Association Colloids

