- The reaction rates vary with the concentration of the reactants.
- The experimental results show that the reaction rates do not always show similar concentration-dependences.
- In many cases, the rates are found to be proportional to the first power of concentration.
- There are many reactions in which the rates varied to the second or even third power of the reactant concentration.
- For a general reaction,
aA + bB + cC → Products
- The reaction rate is described by the expression
Rate = k [A]p[B]q[C]r…………..(i)
where, k is the rate constant of a reaction, [A], [B] and [C] are the molar concentrations of the reactants A, B and C.
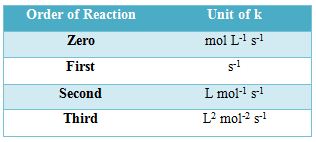
The exponents (p, q and r) are called order of the reaction with respect to A, B and C respectively.
The sum p + q + r is called as the overall order of the reaction.
- Thus, the order of the reaction is equal to the sum powers of all the concentration terms in the rate law equation.
- In other words, the order may be defined as: The total number of concentration variables which determine the rate of any reaction is called the overall order of the reaction.
- The order of a reaction is generally a small integer, half integer or zero.
- The order of the reaction is obtained experimentally.
- As a matter of general rule, reaction order cannot be deduced from the stoichiometry of the reaction.
- However, if the reaction takes place in a single step, then the stoichiometric coefficient(s) of the reactant(s) in the balanced chemical equation give the order of the reaction with respect to the respective reactant.
- The overall order is then given by the sum of all the stoichiometric coefficients.
A) Zero Order Reactions
- The reaction whose rate does not depend upon the concentration of the reactant is called a zero order reaction.
- Thus, the reaction A→ Products is called a zero order reaction if
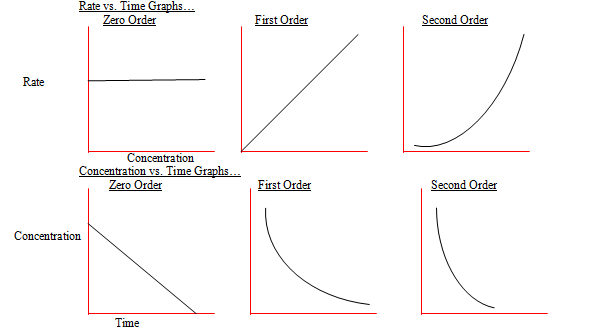
Image Source: Reddit
Rate = -d[A]/dt = k[A]0 = k……………………….(ii)
- Thus, the rate of a zero order reaction remains constant with time.
- Reaction of H2 and Cl2 over the surface of water is a zero order reaction.
B) First Order Reactions
- The reaction for which the rate depends upon only one concentration variable is called the first order reaction.
- The reaction, A → Products is said to be first order, if the reaction rate is given by the equation,
Reaction rate = k[A]
- Thus, the reaction rate in such case changes linearly with the concentration of A (the reactant) i.e., if [A] is doubled, the reaction rate also gets doubled.
- This reaction is then said to be of first order i.e., order of the reaction is one.
- The reaction, aA + bB→ cC + dD, is said to be of first order, if the reaction rate is given by any of the following expressions.
Rt = k[A] or Rt= k[B]
- The reaction rate in such a case would vary linearly with the concentration of A or B.

C) Second Order Reactions
- The reaction in which two concentration variables affect the rate is called a second order reaction.
- The reaction, A → Products, is said to be of second order, if the reaction rate follows the following equation, Reaction rate = k[A]2
- In such a case, the reaction rate varies to the second power of the concentration of the reactant A.
- Thus, for a second order reaction if the concentration of the reactant (A) is doubled, then the reaction rate gets increased by a factor of four.
- The reaction, aA + bB → Products is said to be second order, if the rate of reaction depends upon two concentration variables.
![What is the significance of the kink in the [A] vs T graph for a 2nd order reaction? - Chemistry Stack Exchange](https://i.stack.imgur.com/4SS05.png)
- In such a reaction, the following cases are possible:
| Reactant involved
|
Rate law
|
Order of reaction | ||
| w.r.t A | w.r.t B | overall | ||
| Only A | Rate = k[A]2 | 2 | 0 | 2 |
| Only B | Rate = k’[B]2 | 0 | 2 | 2 |
| Both A and B | Rate = k”[A][B] | 1 |
1 |
2 |
- Some examples of second order reaction are:
2 NO2 → 2 NO + O2 [Nitrogen dioxide decomposes into nitrogen monoxide and an oxygen molecule.]
2 HI → I2 + H2 [Hydrogen Iodide decomposes into iodine gas and hydrogen gas].
2 NOBr → 2 NO + Br2 [In the gas phase, Nitrosyl bromide decomposes into nitrogen oxide and bromine gas.]
References:
i) https://brilliant.org/wiki/order-of-chemical-reactions/
Order of Reaction

