- Elemental oxygen exists as a triatomic molecule (O3) known as ozone in addition to di-oxygen.
- The existence of an element in more than one modification in the same physical state is referred to as allotropy.
- Ozone is an allotropic form of oxygen which is present in the upper atmosphere about 20 km above the surface of the earth.
Preparation of Ozone
- Ozone can be conveniently prepared by passing silent electric discharge through pure, cold and dry oxygen.
- The apparatus used for this purpose is called ozoniser.
3O2 → 2O3; ∆ H= +284 KJ
- The reaction is endothermic and energy is absorbed.
- The silent (sparkles) electric discharge produces less heat. This prevents the local rise in temperature which may decompose ozone back to oxygen.
- Two types of ozonisers that are commonly used for the production of ozone are:
a)Siemen’s ozonizer
b)Brodie’s ozoniser
A) Siemen’s ozoniser
- It consists of two concentric metal tubes sealed together at one end.
- The inner side of the inner tube and outer side of outer tube is coated with tin foil and connected to one terminal each of an induction coil.
- A slow stream of pure and dry oxygen is passed through the annular space and is subjected to the silent electric discharge.
- As a result, 10-15% ozone is formed.
- It comes out along with oxygen called ‘ozonized oxygen’.

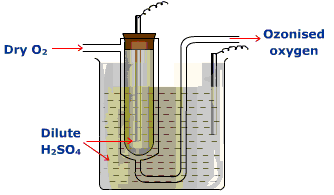
Image source: chemistrylibre
B) Brodie’s ozoniser
- The principle of this ozonizer is similar to Siemen’s ozoniser.
- Two platinum electrodes are dipped in the sulphuric acid and connected to an induction coil.
- A current of pure dry oxygen is passed through the annular space and subjected to silent electric discharge.
- As a result, 15-20% of ozone is formed.
- The ozone so formed is along with oxygen called ‘ozonized oxygen’.
- Ozonized oxygen can be converted to pure ozone by lowering the temperature of the ozone condenses at -112.4oC into liquid while oxygen condenses at -182.5oC.
- Hence, ozone and oxygen can be separated in pure state.

Image source: ozonesecreteriat
Uses of Ozone
- It is used as germicide and disinfectant. So, it finds use in sterilization of water.
- It is used to purify the atmosphere in crowded places like cinema halls, heavy traffic stations and in underground tunnels and mines.

Image source: researchgate
- It is also used as bleaching agent for fabrics, oils, ivory, wax, etc.
- It is also used in laboratory to prepare potassium permanganate and to locate the double bond in organic compounds.
- It is used in aquaculture and fish farming.
- It is used for surface treatment such as semiconductor manufacture, inorganics production.
- It is used for food processing like rinsing water, food preservation.
References:
ii) https://chemicalnote.com/ozone-preparation-uses-test-ozone-layer-depletion/

