- Metal ions can complex with a variety of molecular species to form coordination compounds/ complex ions.
- These coordination compounds/complex ions differ appreciably from each other and also from their constituent species.
- These aspects of coordination compounds/complex ions have been exploited in many ways.
- Some of the major applications of the coordination compounds are described below.
i) In analytical chemistry
- Formation of coordination compounds/complex ions has important role in analytical chemistry.
- It is used for either separating or estimating only one particular ion in the presence of other ions.
- This is done by converting all ions except the one into stable complex ions by adding a suitable reagent (acting as ligand) into the solution.
- This reagent forms stable complexes with all the ions except the desired one.
- This desired ion thus remains as free ion in the solution.
- These complex ions would no longer give the chemical reactions of the constituent ions.
- So, the solution will give test/reactions only of the ion left un-complexed in the solution.
- The ion which exists in the free ionic form in the solution then can be estimated or separated by suitable chemical method.
- This application is illustrated by taking a solution containing both Cu2+ and Cd2+ions.
- To estimate or separate one of these ions, we generally proceed as follows:
a) To the solution containing Cu2+ and Cd2+ ions, an excess of KCN solution is added.
b) KCN dissociates in the solution to give CN– ions which form cyano complexes with both the ions, i.e.,[Cu(CN)4]2- and [Cd(CN)4]2-.
c) On passing H2S through the solution, cadmium gets precipitated as cadmium sulphide (CdS) despite its larger solubility.
Explanation
When KCN is added to a solution containing Cu2+ and Cd2+, both the ions get complexed to form [Cu(CN)4]2- and [Cd(CN)4]2- complex ions in accordance with the reactions,
Cu2+ + 4CN– ⇌ [Cu (CN)4]2-
Cd2+ + 4CN- ⇌ [Cd (CN)4]2-
The complex [Cu (CN)4]2- is more stable than the complex [Cd(CN)4]2–. It means that the equilibrium concentration of free Cd2+ in the solution is much higher than that of free Cu2+. So, when H2S is passed through the solution only CdS gets precipitated.
- Nickel can be separated/identified / estimated by forming its complex with dimethylglyoxime. Nickel dimethylglyoixme is pink in colour.
ii) For the estimation of hardness of water
- Hardness of water is confirmed if there is the presence of Ca2+ and Mg2+ ions in water.
- There is formation of stable complexes with Ca2+ and Mg2+ by EDTA. So, estimation of the total hardness of water can be done by simple volumetric titration of water with EDTA.
- Since, the stability constants of Ca2+ EDTA and Mg2+ EDTA complexes is different, hence the selective estimation of the two ions can be done.
iii) For softening of water
- Ca2+ and Mg2+ ions which cause hardness can form stable complexes with EDTA and polyphosphates.
- So, Ca2+ and Mg2+ ions present in water get trapped by these reagents by complexation which makes the water soft.
iv) In electroplating
- There are various reasons of utilizing formation of complex ions in electroplating:
a) Complexes are more stable than the simple ions in solution towards oxidation, reduction, hydrolysis, etc.
b) Complexes give a controlled supply of metal ions in the solution. It is because, the complexes are reasonably stable, and also they dissociate to very small extent. As a result, the concentration of free metal ions is maintained at low level in solution. This low concentration of metal ions in solution gives better deposit during electroplating.
The complexes commonly used in electroplating are:
K[Ag(CN)2] for silver plating and K[Au(CN)2] for gold plating.
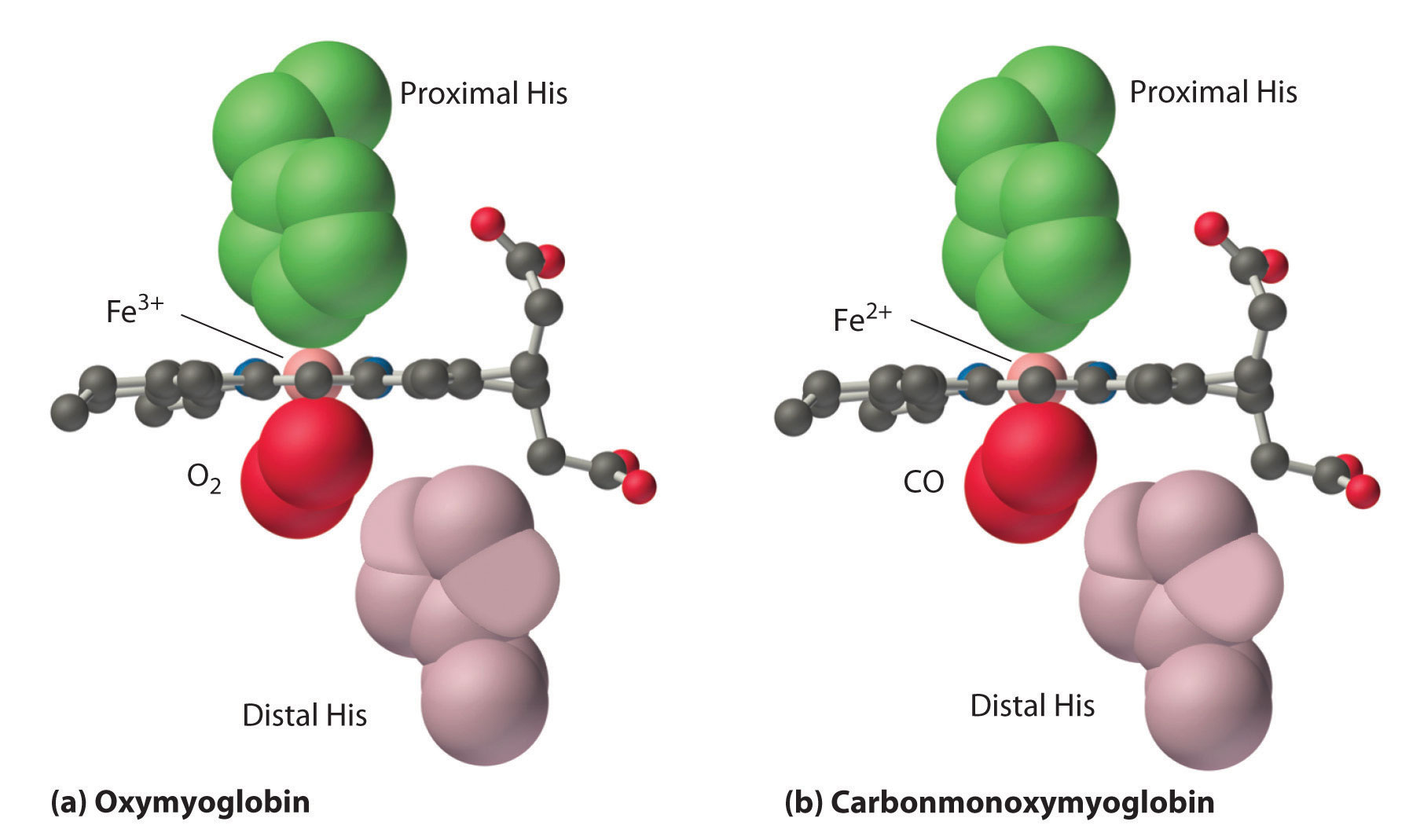
Image Source: chemlibretext
v) In extractive metallurgy
- The usefulness of complex formation is observed in the extraction of metals like the extraction of silver from Ag2S by complexing Ag+ with CN– in accordance with the reaction.
Ag2S + 4NaCN ⇌2Na[Ag(CN)2] + Na2S
- The metallic silver is recovered by treating the solution of Na[Ag(CN)2] with zinc dust.
2Na[Ag(CN)2] + Zn → Na2[Zn(CN)4] + 2Ag
- There are however many more interesting application of complex formation in the field of extractive metallurgy.
vi) In the field of medicine
- Complexes have been found useful in the treatment of certain diseases. For example: the platinum complex, cis-[PtCl2(NH3)2] known as cis-platin is found useful in cancer chemotherapy.

Image Source: Amrita
vii) In nature
- Chelates are special type of complexes. Formation of chelates in living systems has hot much biological significance.
- For example: chlorophyll is a complex of Mg2+ with porphyrin, hemoglobin is a complex of Fe2+with porphyrin. Vitamin B12 is a complex of Co2+.
viii) In modifying the redox behavior of metal ions
- Complexation leads to changes in the redox potential of the metal ions.
- This aspect of complexation has been utilized in catalyzing certain biochemical reactions.
References:
ii) https://study.com/academy/lesson/applications-of-coordination-compounds.html

