- The cyclic movement of nitrogen between atmosphere and living organisms or the exchange of nitrogen between atmosphere and living organisms is known as nitrogen cycle.
- It is also one of the gaseous-cycle.
- Nitrogen is the most essential element for almost all forms of life.
- Living organisms need nitrogen atoms to make amino acids, proteins, enzymes, chlorophyll and nucleic acids (DNA and RNA).
- The atmosphere contains very large amount of nitrogen about 78% of the air is nitrogen gas.
- But only a few organisms are able to make use of this source.
- Even green plants cannot use gaseous nitrogen.
- The nitrogen cycle is completed in several steps by the involvement of different kinds of microorganisms. They are as follows:
A) Nitrogen fixation
- Nitrogen gas is composed of two nitrogen atoms that are bonded together by the help of three covalent bonds.
- This arrangement is very stable and so nitrogen molecules are very unreactive.
- Most plants cannot utilize atmospheric nitrogen directly but only the combined or fixed forms such as nitrate (NO3), nitrite (NO2) and ammonium (NH4+), which are more reactive and can take part in metabolic reactions.
- The conversion of atmospheric nitrogen gas to these more reactive substances is called nitrogen fixation.
- The nitrogen fixation basically occurs in two ways. They are:
i) Non- biological fixation of Nitrogen
- Nitrogen fixation occurs whenever lightning flashes through the air.
- The huge amounts of energy in a lightning flash can break nitrogen molecules apart and allow nitrogen atoms to react with oxygen in the air to form nitrogen oxides.
N2 + 2 (O) → 2NO
2NO + 2 (O) → 2NO2
- These nitrogen oxides get dissolved in raindrops and fall to the ground.
- Then, they react with mineral compounds to form nitrites, nitrates and ammonia.
- Very large quantities of nitrogen are fixed industrially in the Haber process, to produce nitrogen, containing fertilizers.
- Nitrogen and hydrogen gas are reacted to each-other under high pressure to produce ammonia, NH3.
- Some of the ammonia is then used to make nitric acid, which in turn can be used to make nitrates.
- Ammonium nitrate (NH4NO3) is the most widely used fertilizer in the world.
ii) Biological fixation of Nitrogen
- Some bacteria and blue green algae are able to make use of the atmosphere’s nitrogen and change it into nitrogenous compounds.
- It occurs by two methods:
a) Non-symbiotic Nitrogen Fixation
- The nitrogen fixation is also carried out by many free living bacteria and blue green algae which utilize molecular nitrogen non-symbiotically.
- The free living nitrogen fixers can be divided into different categories.
- Generally, the N2 fixation is associated with forms containing the heterocyst.
- Heterocyst is large thick walled empty cooking cells.
- They provide favourable condition for the effective functioning of nitrogenase and bring about nitrogen fixation.
- Examples are: Azotobacter, Beijerinckia, Derxia, Clostridium, Bacillus, Enterobacter, Rhodomicrobium, Anabaena, Nostoc, Calothrix, etc.
b)Symbiotic Nitrogen Fixation
- Nitrogen fixation is also known as biological N2 fixation because the major amount of nitrogen is fixed by biological process.
- In biological N2 fixation, major part is covered by nodule forming symbiotic bacteria.
- Major nitrogen fixers in nature are the Rhizobium spp.
- It lives freely in soil and in the root region both with leguminous and non-leguminous plants.
- They enter into symbiosis only with leguminous plants.
- The Rhizobium spp. are gram negative, non-spore forming, motile and aerobic rods having 0.5- 0. 9 µm width and 1.2- 3 µm length.
- They infect the roots and form the nodules.
- In symbiosis, nitrogenous nutrient is added by Rhizobium whereas carbon source is provided by plants.
- On death and decay of host plant, a good amount of nitrogenous matter is made available to the soil.
B) Nitrogen used by plants and animals
- As a result of fixation, atmospheric nitrogen reaches to soil as nitrates, nitrites and ammonia.
- These are absorbed by plants.
- Inside the plant cells, nitrate ions are reduced to ammonium ions which can then be used to make amino acids.
- The plant can then use the amino acids to make proteins, enzymes, nucleic acids, etc.
- Animal obtain their nitrogen in the form of proteins, when they eat plants or other animals.
- In animals it is converted into animal proteins and eventually down into nitrogenous wastes such as urea, uric acid and ammonia and excreted out.

Image source: thebiologynotes
C) Decomposition of organic nitrogenous compounds
- The dead and decaying bodies of plants and animals and excreta of animals are decomposed by bacteria and fungi in the soil and nitrogen is converted back to free nitrogen in following ways:
i) Ammonification
- It is the process in which organic nitrogen compound is converted into inorganic nitrogen compounds.
- In other words, it is the process of release of ammonia.
- It includes two steps. They are: proteolysis and deamination.
- Proteolysis is the degradation of organic nitrogenous compounds into amino acids.
- The animal proteins remain on the soil after death of animals which is similar in case of plants as well.
- The proteins of plants and animals will be decomposed by micro-organisms or micro-organisms themselves are decomposed into soil.
- Some examples of proteolytic micro-organisms are Bacillus, Clostridium, Pseudomonas, Micrococcus, Aspergillus, Alternaria, proteolytic Actinomyctes, etc.
- Deamination is the conversion of amino acid into ammonia which occurs in the end product of proteolysis i.e. amino acid.
- Product of deamination differs with enzyme system of micro-organisms.
- Ammonia can also be absorbed by few plants from the soil.
ii) Nitrification
- The most reduced form of inorganic nitrogen, serves as the starting point for a process known as nitrification.
- It is very important and occurs in two steps.
- It is the oxidation of ammonia into nitrite then into nitrate.
- The importance of nitrifying microorganisms rests to a great degree on their capacity to produce the nitrate that is the major nitrogen source assimilated by higher plants.
- Firstly, ammonia is oxidized to nitrite mainly by Nitrobacter, Nitrocystis, Nitrospira, etc.
2NH3 + 3O2 → 2HNO2 + 2H2O + 16 Kcal.
- Secondly, the nitrite is converted to nitrate by mainly Nitrosomonas and Nitrobacter.
- These two are major nitrifying oblique aerobic chemoautotrophs.
2HNO2 + O2 → 3HNO3 +18 Kcal
- During nitrification, other intermediate products are also formed which are unstable.
- They are: hydroxylamine, hyponitrite which is highly toxic and unstable.
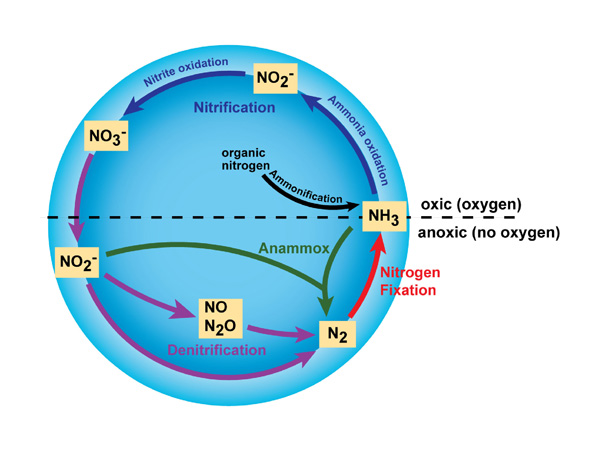
Image source: nature

iii) De-nitrification
- It is the reduction process which involves the microbial reduction of nitrate to nitrite and then to molecular nitrogen.
- Reduction of NO2– and NO3– is also known as de-nitrification.
- This process requires anaerobic condition which can also be of two types.
a) Nitrate respiration
- The de-nitrification process in which nitrate replaces oxygen is known as nitrate respiration which is also an anaerobic respiration process.
b) Nitrate assimilation
- If nitrate is utilized only as a source of nutrient and not as terminal electron acceptor the process is known as nitrate assimilation.
- Bacteria like Pseudomonas, Bacillus, Achromobacter, Micrococcus, Thiobacillus, Vibrio, etc are responsible in this process.
- The biochemical reactions included are:
2NO3– +10H → N2 + 4H2O + 2OH–
2NO2– + 6H → N2 + 2H2O + OH–
N2O + 2H → N2 + H2O
- Nitrobacillus denitrificans which oxidizes sulphur chemoautotrophically also reduces nitrate to nitrogen.
5S + 6KNO3 + H2O → 3N2 + K2SO4 + 4KHSO4
- Anaerobic bacteria like Clostridium may reduce NO3– to NH3
HNO2 + 4H2 → NH3 + 3H2O
References:
i) https://www.sciencelearn.org.nz/resources/960-the-nitrogen-cycle
ii) https://biologydictionary.net/nitrogen-cycle/
iii) https://biologydictionary.net/nitrogen-cycle/
iii) https://www.lenntech.com/nitrogen-cycle.htm

