- Vitamin C is a water soluble versatile vitamin.
- It has important role in human health and disease.
- It has become the most controversial vitamin in the recent years.
- Its controversy is due to the claims and counterclaims on the use of it in mega-doses to cure everything from common cold to cancer.
- The disease associated with this vitamin deficiency is scurvy which has been known for centuries.
- This was the first disease found to be associated with diet.
- Around 10000 mariners died of a miraculous disease due to lack of fresh vegetables in their diet in sixteenth century.
- A surgeon of the English navy named James Lind published ‘Treatise on scurvy’ in 1753.
- From his observations, the Royal Navy started using and supplying lime or lemon to all the crews.
- They used to carry crates of lemons, hence they were also known by Limeys.
Chemistry of Vitamin C
- Ascorbic acid is a hexose (6 carbons) derivative and closely resembles mono-saccharides in structure.
- The enolic hydroxyl group provides it with the acidic property.
- It is a strong reducing agent.
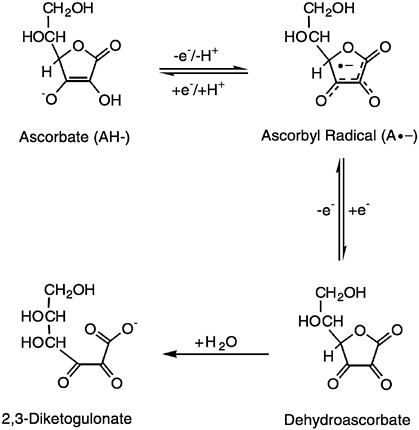
- L-Ascorbic acid undergoes oxidation to form de-hydro-ascorbic acid and this reaction is reversible.
- Both ascorbic and de-hydro-ascorbic acid are biologically active.
- However, D-ascorbic is not active.
- The plasma and tissues predominantly contain ascorbic acid in the reduced form.
- The ratio of ascorbic acid to de-hydro-ascorbic acid in many tissues is 15: 1.
- On hydration, de-hydro-ascorbic acid is irreversibly converted to 2, 3- diketogulonic acid which is inactive.
- Hydration reaction is almost spontaneous, in alkaline or neutral solution.
- So, oxidation of vitamin c is regarded as the biological inactivation.
- Oxidation of ascorbic acid is rapid in the presence of copper.
- For this reason, vitamin C becomes inactive if the foods are prepared in copper vessels.
Biochemical functions
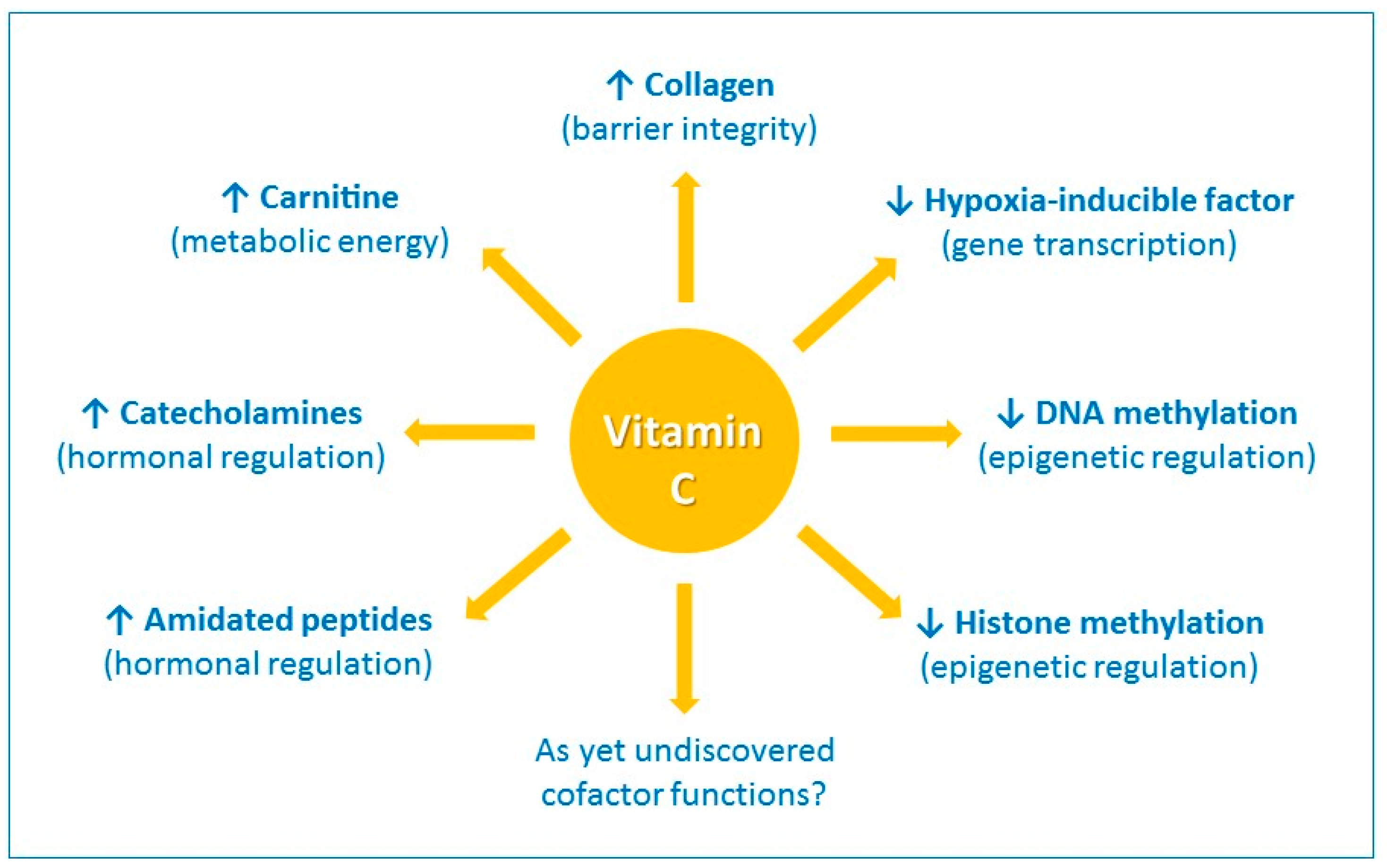
- Vitamin C has got property to undergo reversible oxidation reduction i.e. inter-conversion of ascorbic acid and de-hydro-ascorbic acid.
- The functions of vitamin C exist due to this property. The various functions are:
a)Collagen formation
- It acts as coenzyme in hydroxylation of proline and lysine while proto-collagen is converted to collagen (i.e. post-translational modification).
- Lysyl hydroxylase catalyzes the hydroxylation reaction for lysine and prolyl hydroxylase for proline.
- This reaction is dependent on Vitamin C, molecular oxygen and α- ketoglutarate.
- Hydroxyproline and hydroxylysine are essential for the collagen cross linking and the strength of the fiber.
- Thus, Vitamin C helps in maintaining normal connective tissue and healing the wound.
b)Bone formation
- Vitamin C is required for bone formation as bone tissues possess organic matrix, collagen and the inorganic calcium, phosphate, etc.
c)Iron and hemoglobin metabolism
- Ascorbic acid enhances the iron absorption by keeping it in the ferrous form.
- It is due to the reducing property of Vitamin C.
- It helps in the formation of ferritin (storage form of iron) and mobilization of iron from ferritin.
- It also converts methemoglobin to hemoglobin.
- The formation of bile pigments by degradation of hemoglobin is also due to the ascorbic acid.
d)Tryptophan metabolism
- It is also required for the hydroxylation of tryptophan to hydroxyl-tryptophan in the synthesis of serotonin by enzyme hydroxylase.
e)Tyrosine metabolism
- It also helps in oxidation of p- hydroxyl phenylpyruvate (enzyme- hydroxylase) to homogentisic acid in Tyrosine metabolism.


f)Folic acid metabolism
- The active form of the vitamin folic acid is tetra-hydrofolate (FH4).
- Vitamin C is needed for the formation of FH4 by the help of enzyme folic acid reductase.
- Further, in association with FH4, ascorbic acid is involved in the maturation of erythrocytes.
g)Peptide hormone synthesis
- Many peptide hormones contain carboxyl terminal amide which is derived from terminal glycine.
- Hydroxylation of glycine is carried out by peptidylglycine hydroxylase which requires vitamin c.
h)Synthesis of corticosteroid hormones
- Adrenal gland possesses high levels of ascorbic acid particularly in the periods of stress.
- It is believed that Vitamin C is required for the hydroxylation reactions in the synthesis of corticosteroid hormones.
i)Sparing action of other vitamins
- Ascorbic acid is a strong antioxidant which spares vitamin A, vitamin E, and some B-complex vitamins from oxidation.
j)Immunological function
- Vitamin C enhances the synthesis of immunoglobulins (antibodies) and increases the phagocytic action of leukocytes.
k)Preventive action on cataract
l)Preventive action on chronic diseases like cancer, cataract and coronary heart diseases.
Recommended dietary allowance (RDA)
- About 60-70 mg Vitamin C intake per day will meet the adult requirement.
- Additional intakes (20-40% increase) are recommended for women during pregnancy and lactation.
Dietary sources
- Citrus fruits, gooseberry (amla), guava, green vegetables (cabbage, spinach), tomatoes, potatoes (particularly skin) are rich in ascorbic acid.
- High content of vitamin c is found in adrenal gland and gonads.
- Milk is a poor source of ascorbic acid.
Deficiency symptoms
- Its deficiency results in scurvy.
- This disease is characterized by spongy and sore gums, loose teeth, anemia, swollen joints, fragile blood vessels, decreased immune-competence, delayed wound healing, sluggish hormonal function of adrenal cortex and gonads, hemorrhage, osteoporosis, etc.
- Most of these symptoms are related to impairment in the synthesis of collagen and the antioxidant property of the vitamin C.
References:
i) https://www.hsph.harvard.edu/nutritionsource/vitamin-c/
ii) https://www.nhs.uk/conditions/vitamins-and-minerals/vitamin-c/

