- A voltaic cell is an electrochemical cell used to produce electrical energy with the help of a chemical reaction.
- It is the primary cell which was designated by Alessandro Volta in 1800.
Construction
- A simple construction of a voltaic cell is shown in figure below.
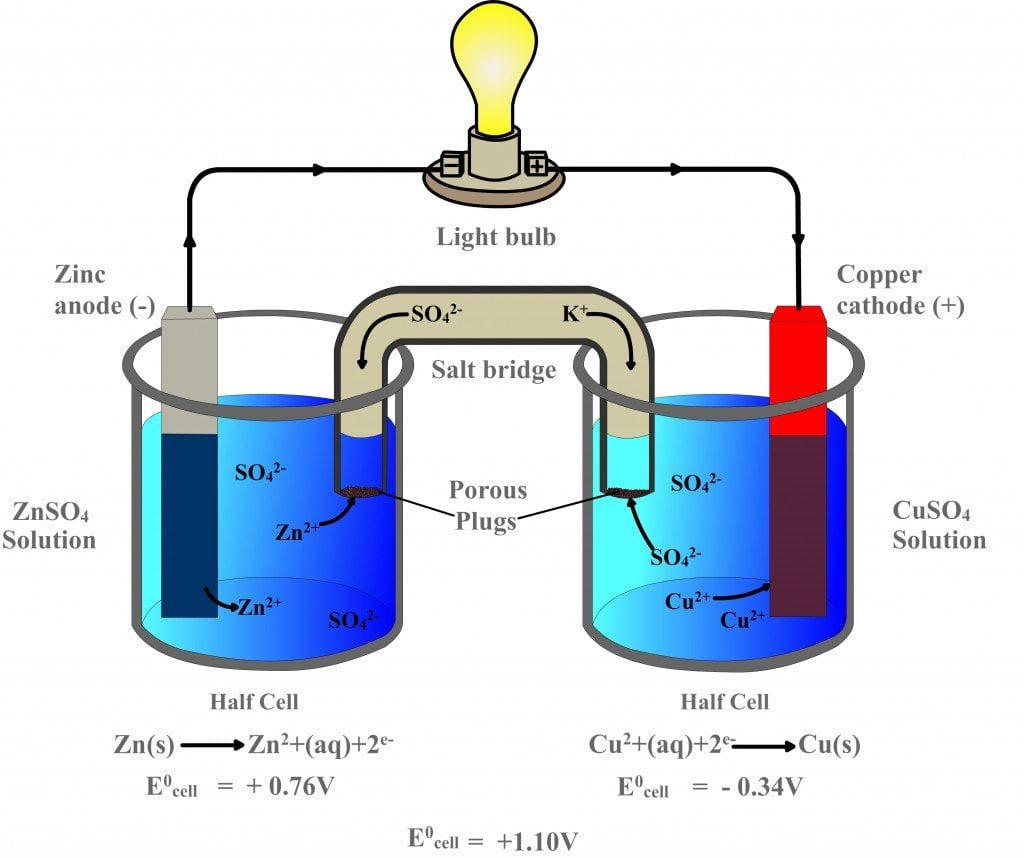
- It consists of a copper plate and a zinc plate immersed in a dilute sulphuric acid (H2SO4) contained in a glass vessel.
- The copper plate acts as an anode and the zinc plate acts as cathode. The H2SO4 acts as an electrolyte.
Action of cell
Sulphuric acid is dissociated into ions as: H2SO4 →2H+ + SO4—
At Cathode
- Initially, the zinc atoms are neutral. As soon as the zinc plate is placed in H2SO4, the zinc atoms react with the sulphate ions.
- In other words, a zinc atom goes into the solution as zinc ion (Zn++) leaving behind two electrons (2e–) at the cathode.
- The reaction is: Zn → Zn2+ + 2e–
- The Zn++ ion reacts with SO4— ion to form zinc sulphate as: Zn++ + SO4— → ZnSO4
- The result is that the zinc atoms go into the solutions as ZnSO4 and the zinc plate is negatively charged. This means that the zinc plate is consumed.
At Anode
- When the concentration of Zn++ ion near the zinc plate becomes high, the H+ ions in the solution are repelled towards the copper plate.
- When the hydrogen ions reach the copper plate, they remove electrons from the copper atoms and become hydrogen gas.
- The reaction is : 2H+ + 2e–→ Cu++ + 2e–
- Hence, the copper plate is positively charged.
- The action at the cathode continues until the negative charges collected in it are good enough to stop the further ionization of the zinc atoms of the zinc plate.
- Similarly, the action at the anode continues until the positive charges in it are good enough to stop the further movement of the hydrogen ions in the solution towards the copper plate due to Zn++ ion concentration near the anode.
- When the equilibrium is established, the copper plate has a collection of electrons.
- The amount of positive charge in the copper plate is equal to the amount of negative charge in the zinc plate.
- Due to the positive charge, the copper plate is at positive potential and hence it acts as anode.
- Due to the negative charge, the zinc plate is at negative potential and hence acts as cathode.
- Thus, a difference is established between the copper and the zinc plate and hence the cell acts as a source of e.m.f.
- If the copper plate and zinc plate are connected to an external circuit, the electrons flow from the zinc plate to the copper plate.
- When two electrons reach the copper plate, they neutralize Cu++ ion.
- Hence, the equilibrium between the charges in the plates is disturbed. The potential difference across the plates decreases.
- To recover the original potential difference, more zinc atoms go into the solution as ions and so on.
- In this way, the zinc plate goes on behind used up. The cell stops delivering current when all the zinc is dissolved into the solution or all sulphuric acid is consumed.
- The e.m.f of voltaic cell is about 1.1 volt.
Defects of Voltaic Cell
A voltaic cell suffers from two types of defects. They are as follows:
i) Local action
- If commercial zinc is used in zinc plate, it contains impurities like iron, cobalt, lead, etc.
- If these impurities are present, the impurity atoms form small short circuited cells with the zinc atoms.
- Such short circuited cells formed between the zinc atoms and the impurity atoms go on consuming the zinc plate even if the cell is not delivering current to external circuit.
- This defect of voltaic cell is called local action.
Remedy
- The local action can be removed by using chemically pure zinc or amalgamated zinc.
- The amalgamated zinc is the zinc rubbed with mercury.
- In doing so, the impurity atoms if present are covered by the mercury atoms.
- As a result, the local action cannot take place. The mercury atoms do not avoid the zinc atoms from coming into the solution.
ii) Polarization
- At the copper plate of voltaic cell, hydrogen is liberated in the form of gas bubbles.
- Some of the bubbles form a layer on the copper plate. When the hydrogen bubbles form a layer around the copper plate, it has the following effects:
- The internal resistance of the cell increases since the air is a bad conductor of electricity.
- The hydrogen bubbles prevent the hydrogen ions from reaching the copper plate. If the hydrogen ions do not reach the copper plate, they form a layer of positive ion in the cell. The layer of hydrogen ions and the zinc plate establish e.m.f called back e.m.f which reduces the e.m.f of the cell. This defect of a voltaic cell is called polarization.
Remedy
This defect can be removed by the following ways:
- By brushing the copper plate time to time thus removing the hydrogen bubbles, or
- By using an oxidizing agent which reacts with the hydrogen to form water. The oxidizing agent is called depolarizer.
References:
ii) https://byjus.com/chemistry/galvanic-cell/
iii) https://www.thoughtco.com/galvanic-cell-definition-604080

