A) The electronic configuration(EC)
- The EC of alkali metal is ns1.
- They have single electron in their outer most shell.
- Therefore, their valency is also one.
- They have a great tendency of losing electron from their outermost orbit.
- Due to the above reason, they are considered to be the most electropositive elements.
- For example: Na (11) =1s22s22p63s1
- As the last electron is filled in s subshell, they are called s-block elements.
B) Density
- It is quite low in alkali metals.
- It increases down the group from Li to Cs due to increase in their atomic mass which compensates more than its bigger atomic size.
- However, K is lighter than Na which is due to abnormal increase in its atomic size.
- Thus, density follows the order as:
Li< K< Na< Rb< Cs< Fr
C) Melting point (m.p) and boiling point (b.p)
- The m.p and b.p of these metals are very low.
- They decrease further with increase in atomic number due to weakening of metallic bond.
- Fr is liquid at room temperature.
D) Softness
- They are silvery white and soft wax and can be cut with knife.
E) Ionization energy (IE)
- IE are very low due to presence of only one electron in outermost orbit.
- They more often prefer to lose ns1 electron to change to M+
- IE decreases down the group from Li to Fr due to increase in atomic radius and decrease in nuclear attraction.
Element Li Na K Rb Cs Fr
IE1 (KJ mol-1) 520 496 419 403 376 400
F) Flame colouration
- Alkali metals and their salts impart characteristics colour to their flame.
- It is due to the release of excitation energy in the visible region.
- The energy is absorbed by them which is released when heated in a bunsen flame.
Li Na K Rb Cs
Crimson red golden yellow violet violet violet
G) Electropositive character
- They are strongly electro positive.
- It is due to their low ionization energies.
- They form uni-positive ions.
H) Atomic radii
- Atomic radii are largest in their respective periods.
- They increase down the group due to increase in principal shells.
I) Ionic radii
- Ionic radii of these metals are much smaller than their corresponding atomic radii.
- It is due to lesser number of shells and contractive effect of increased nuclear charge.
J) Photoelectric effect
- K and Na especially show photoelectric effect.
- It is due to low ionization energies.
(Photoelectric effect – eject electrons when exposed to light).
K) Electrical conductivity
- They are good conductors of heat and electricity.
- Due to low ionization energies, their valence s-electrons are loosely held by the metal ions in solid state.
- Electrical conductivity increases down the group.
- It is due to the decrease in the ionization energy in the order as
Li+< K+< Na+ < Rb+< Cs+
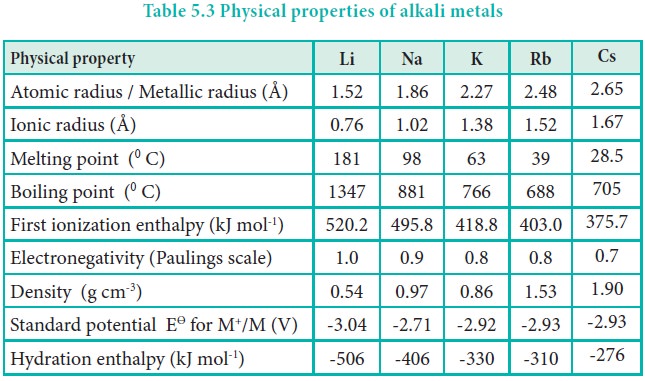
Image source: braincart
L) Reducing character
- All alkali metals are good reducing agents.
- It is due to their low ionization energies.
- There reducing character follows the order:
Na <K< Rb< Cs< Li
- The reducing character of metals depends upon a) heat of vaporization b) ionization energy and c) heat of hydration.
- Since, Li+ ion has the smallest size, its heat of hydration is highest.
- Therefore among alkali metals Li has the highest negative electrode potential and is the strongest reducing agent.
M) Crystal structure
- All alkali metals possess body centered cubic structures with coordination number 8.
References:
i) https://study.com/academy/lesson/alkali-metal-elements-properties-characteristics-reactions.html
ii) https://www.thoughtco.com/alkali-metals-606645
Trends in atomic and physical properties of alkali metals

